Implications of bipolar voltage mapping and magnetic resonance imaging resolution in biventricular scar characterization after myocardial infarction
- PMID: 30239689
- PMCID: PMC6321957
- DOI: 10.1093/europace/euy192
Implications of bipolar voltage mapping and magnetic resonance imaging resolution in biventricular scar characterization after myocardial infarction
Abstract
Aims: We aimed to study the differences in biventricular scar characterization using bipolar voltage mapping compared with state-of-the-art in vivo delayed gadolinium-enhanced cardiac magnetic resonance (LGE-CMR) imaging and ex vivo T1 mapping.
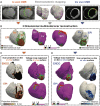
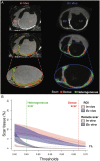
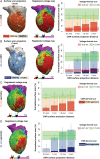
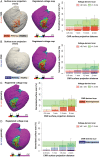
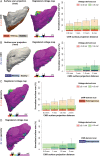
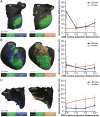
Methods and results: Ten pigs with established myocardial infarction (MI) underwent in vivo scar characterization using LGE-CMR imaging and high-density voltage mapping of both ventricles using a 3.5-mm tip catheter. Ex vivo post-contrast T1 mapping provided a high-resolution reference. Voltage maps were registered onto the left and right ventricular (LV and RV) endocardium, and epicardium of CMR-based geometries to compare voltage-derived scars with surface-projected 3D scars. Voltage-derived scar tissue of the LV endocardium and the epicardium resembled surface projections of 3D in vivo and ex vivo CMR-derived scars using 1-mm of surface projection distance. The thinner wall of the RV was especially sensitive to lower resolution in vivo LGE-CMR images, in which differences between normalized low bipolar voltage areas and CMR-derived scar areas did not decrease below a median of 8.84% [interquartile range (IQR) (3.58, 12.70%)]. Overall, voltage-derived scars and surface scar projections from in vivo LGE-CMR sequences showed larger normalized scar areas than high-resolution ex vivo images [12.87% (4.59, 27.15%), 18.51% (11.25, 24.61%), and 9.30% (3.84, 19.59%), respectively], despite having used optimized surface projection distances. Importantly, 43.02% (36.54, 48.72%) of voltage-derived scar areas from the LV endocardium were classified as non-enhanced healthy myocardium using ex vivo CMR imaging.
Conclusion: In vivo LGE-CMR sequences and high-density voltage mapping using a conventional linear catheter fail to provide accurate characterization of post-MI scar, limiting the specificity of voltage-based strategies and imaging-guided procedures.
Figures
References
-
- Andreu D, Penela D, Acosta J, Fernandez-Armenta J, Perea RJ, Soto-Iglesias D. et al. Cardiac magnetic resonance-aided scar dechanneling: influence on acute and long-term outcomes. Heart Rhythm 2017;14:1121.. - PubMed
-
- Nuhrich JM, Kaiser L, Akbulak RO, Schaffer BN, Eickholt C, Schwarzl M. et al. Substrate characterization and catheter ablation in patients with scar-related ventricular tachycardia using ultra high-density 3-D mapping. J Cardiovasc Electrophysiol 2017;28:1058–67. - PubMed
-
- Amado LC, Gerber BL, Gupta SN, Rettmann DW, Szarf G, Schock R. et al. Accurate and objective infarct sizing by contrast-enhanced magnetic resonance imaging in a canine myocardial infarction model. J Am Coll Cardiol 2004;44:2383–9. - PubMed
-
- Santangeli P, Marchlinski FE.. Substrate mapping for unstable ventricular tachycardia. Heart Rhythm 2016;13:569–83. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

