Recent Advances in Synthesis of 4-Arylcoumarins
- PMID: 30241375
- PMCID: PMC6222890
- DOI: 10.3390/molecules23102417
Recent Advances in Synthesis of 4-Arylcoumarins
Abstract
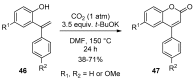
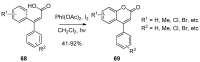
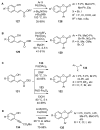
4-Arylcoumarins (4-aryl-2H-1-benzopyran-2-one), also known as neoflavones, comprise a minor subclass of naturally occurring flavonoids. Because of their broad-spectrum biological activities, arylcoumarins have been attracting the attention of the organic and medicinal chemistry communities, and are considered as an important privileged scaffold. Since the development of Pechmann condensation, a classical acid-catalyzed condensation between phenol and β-keto-carboxylic acid, several versatile and efficient synthetic approaches for 4-arylcoumarins have been reported. This review summarizes recent advances in the synthesis of the 4-arylcoumarin scaffold by classifying them based on the final bond-formation type. In particular, synthetic methods executed under mild and highly efficient conditions, such as solvent-free reactions and transition metal catalysis, are highlighted.
Keywords: 4-arylcoumarins; Pechmann condensation; Wittig-type olefination; aldol-type olefination; cyclocarbonylation; cycloisomerization; hydroarylation; transition-metal-catalysis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures










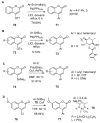
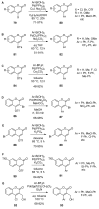

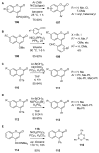

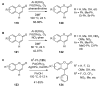
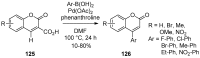
References
-
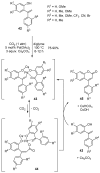
- Garazd M.M., Garazd Y.L., Khilya V.P. Neoflavones. 2. Methods for synthesizing and modifying 4-arylcoumarins. Chem. Nat. Comp. 2005;41:245–271. doi: 10.1007/s10600-005-0126-7. - DOI
-
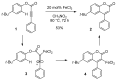
- Wu J., Wang L., Fathi R., Yang Z. Palladium-catalyzed cross-coupling reactions of 4-tosylcoumarin and arylboronic acids: Synthesis of 4-arylcoumarin compounds. Tetrahedron Lett. 2002;43:4395–4397. doi: 10.1016/S0040-4039(02)00718-9. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

