Validation of the 21-gene test as a predictor of clinical response to neoadjuvant hormonal therapy for ER+, HER2-negative breast cancer: the TransNEOS study
- PMID: 30242578
- PMCID: PMC6394785
- DOI: 10.1007/s10549-018-4964-y
Validation of the 21-gene test as a predictor of clinical response to neoadjuvant hormonal therapy for ER+, HER2-negative breast cancer: the TransNEOS study
Abstract
Purpose: The Recurrence Score test is validated to predict benefit of adjuvant chemotherapy. TransNEOS, a translational study of New Primary Endocrine-therapy Origination Study (NEOS), evaluated whether Recurrence Score results can predict clinical response to neoadjuvant letrozole.
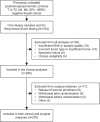
Methods: NEOS is a phase 3 clinical trial of hormonal therapy ± adjuvant chemotherapy for postmenopausal patients with ER+, HER2-negative, clinically node-negative breast cancer, after six months of neoadjuvant letrozole and breast surgery. TransNEOS patients had tumors ≥ 2 cm and archived core-biopsy samples taken before neoadjuvant letrozole and subsequently sent for Recurrence Score testing. The primary endpoint was to evaluate clinical (complete or partial) response to neoadjuvant letrozole for RS < 18 versus RS ≥ 31. Secondary endpoints included evaluation of clinical response and rate of breast-conserving surgery (BCS) by continuous Recurrence Score result, ESR1 and PGR single-gene scores, and ER gene-group score.
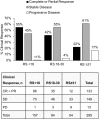
Results: Of 295 TransNEOS patients (median age 63 years; median tumor size 25 mm; 66% grade 1), 53.2% had RS < 18, 28.5% had RS18-30, and 18.3% had RS ≥ 31. Clinical response rates were 54% (RS < 18), 42% (RS18-30), and 22% (RS ≥ 31). A higher proportion of patients with RS < 18 had clinical responses (p < 0.001 vs. RS ≥ 31). In multivariable analyses, continuous Recurrence Score result (p < 0.001), ESR1 score (p = 0.049), PGR score (p < 0.001), and ER gene-group score (p < 0.001) were associated with clinical response. Recurrence Score group was significantly associated with rate of BCS after neoadjuvant treatment (RS < 18 vs. RS ≥ 31, p = 0.010).
Conclusion: The Recurrence Score test is validated to predict clinical response to neoadjuvant letrozole in postmenopausal patients with ER+, HER2-negative, clinically node-negative breast cancer.
Keywords: Breast cancer; Hormonal therapy; Neoadjuvant; Oncotype DX; Recurrence score.
Conflict of interest statement
Conflict of interest
Hiroji Iwata: Remuneration: AstraZeneca, Chugai, Daiichi-Sankyo, Eisai, Genomic Health, Novartis, Pfizer, Taiho. Consultant/advisory role: AstraZeneca, Chugai, Daiichi-Sankyo, Kyowa Hakko Kirin, Lilly, Novartis, Pfizer. Funding: AstraZeneca, Bayer, Chugai, Daiichi-Sankyo, Eisai, GSK, Kyowa Hakko Kirin, Lily, MSD, Novartis, Pfizer. Other: CSPOR BC organizing committee member. Norikazu Masuda: Remuneration: AstraZeneca, Chugai, Pfizer, Takeda. Funding to institution: AstraZeneca, Chugai, Daiichi-Sankyo, Kyowa Hakko Kirin, Lilly, MSD, Novartis, Pfizer. Yutaka Yamamoto: Remuneration: AstraZeneca, Chugai, Daiichi-Sankyo, Eisai, Genomic Health, Kyowa Hakko Kirin, Novartis, Pfizer, Taiho, Takeda. Funding: AstraZeneca, Chugai, Novartis, Taiho. Tatsuya Toyama: Remuneration: AstraZeneca, Chugai, Daiichi-Sankyo, Eisai, Ono Pharmaceutical, Taiho, Takeda. Funding: AstraZeneca, Chugai, Daiichi-Sankyo, Eisai, Kyowa Hakko Kirin, Nippon Kiyaku, Novartis, Pfizer, Taiho, Takeda. Masahiro Kashiwaba: Remuneration: AstraZeneca, Chugai, Kyowa Hakko Kirin, Novartis, Pfizer. Shoichiro Ohtani: Remuneration: Chugai, Eisai. Tomomi Fujisawa, Naruto Taira, Takehiko Sakai, Yoshie Hasegawa, Rikiya Nakamura, Hiromitsu Akabane, Yukiko Shibahara, and Hironobu Sasano: None. Takuhiro Yamaguchi: Remuneration: Daiichi-Sankyo. Consulting or advisory role: CAC Croit Corporation, Chugai, Daiichi-Sankyo, Japan Tobacco, Kowa Company, Ono Pharmaceutical, Tsumura & Co. Funding: A2 Healthcare Corporation, AC Medical Inc., CAC Croit Corporation, Daiichi-Sankyo, FMD K&L Japan K.K., Japan Media Corporation, Japan Tobacco Inc., Kyowa Hakko Kirin, Luminary Medical K.K., Medidata Solutions, Inc., Ono Pharmaceutical. Kentaro Sakamaki: Remuneration: Chugai, Novartis, Taiho. Travel expenses: Genomic Health. Helen Bailey: Employment and stock ownership: Genomic Health. Diana B. Cherbavaz: Stock ownership: Genomic Health. Debbie M. Jakubowski: Employment and stock ownership: Genomic Health. Naoko Sugiyama: Employment and stock ownership: Genomic Health. Calvin Chao: Employment and stock ownership: Genomic Health. Yasuo Ohashi: Remuneration: Daiichi-Sankyo, Eisai, Public Health Research Foundation, Sanofi. Research grant: Eisai. Consultant/advisory role: Chugai, Kowa, Taiho. Travel expenses: Takeda, Yakult Honsha. Other: Shionogi, Statcom.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was reviewed and approved by each institution’s review board and/or ethics committee.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Figures
References
-
- Fisher B, Brown A, Mamounas E, Wieand S, Robidoux A, Margolese RG, Cruz AB, Jr, Fisher ER, Wickerham DL, Wolmark N, DeCillis A, Hoehn JL, Lees AW, Dimitrov NV. Effect of preoperative chemotherapy on local-regional disease in women with operable breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-18. J Clin Oncol. 1997;15(7):2483–2493. doi: 10.1200/JCO.1997.15.7.2483. - DOI - PubMed
-
- Mauriac L, MacGrogan G, Avril A, Durand M, Floquet A, Debled M, Dilhuydy JM, Bonichon F. Neoadjuvant chemotherapy for operable breast carcinoma larger than 3 cm: a unicentre randomized trial with a 124-month median follow-up. Institut Bergonie Bordeaux Groupe Sein (IBBGS) Ann Oncol. 1999;10(1):47–52. doi: 10.1023/A:1008337009350. - DOI - PubMed
-
- Semiglazov VF, Semiglazov VV, Dashyan GA, Ziltsova EK, Ivanov VG, Bozhok AA, Melnikova OA, Paltuev RM, Kletzel A, Berstein LM. Phase 2 randomized trial of primary endocrine therapy versus chemotherapy in postmenopausal patients with estrogen receptor-positive breast cancer. Cancer. 2007;110(2):244–254. doi: 10.1002/cncr.22789. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous