Human embryonic stem cell-derived retinal pigment epithelium transplants as a potential treatment for wet age-related macular degeneration
- PMID: 30245845
- PMCID: PMC6143607
- DOI: 10.1038/s41421-018-0053-y
Human embryonic stem cell-derived retinal pigment epithelium transplants as a potential treatment for wet age-related macular degeneration
Abstract
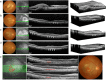
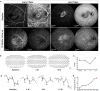
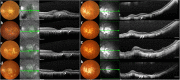
Stem cell therapy may provide a safe and promising treatment for retinal diseases. Wet age-related macular degeneration (wet-AMD) is a leading cause of blindness in China. We developed a clinical-grade human embryonic stem cell (hESC) line, Q-CTS-hESC-2, under xeno-free conditions that differentiated into retinal pigment epithelial cells (Q-CTS-hESC-2-RPE). A clinical trial with three wet-AMD patients was initiated in order to study the safety and tolerance to Q-CTS-hESC-2-RPE cell transplants. The choroidal neovascularization membrane was removed and then a suspension of 1 × 106 Q-CTS-hESC-2-RPE cells were injected into a subfoveal pocket. The patients were followed for 12 months during which no adverse effects resulting from the transplant were observed. Anatomical evidence suggested the existence of new RPE-like cell layer in the previously damaged area. Visual and physiological testing indicated limited functional improvement, albeit to different degrees between patients. This study provides some promising early results concerning the use of transplanted hESC-RPE cells to alleviate wet-AMD.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

