AIDS Therapy Evaluation in the Netherlands (ATHENA) national observational HIV cohort: cohort profile
- PMID: 30249631
- PMCID: PMC6169757
- DOI: 10.1136/bmjopen-2018-022516
AIDS Therapy Evaluation in the Netherlands (ATHENA) national observational HIV cohort: cohort profile
Abstract
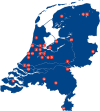
Purpose: In 1998, the AIDS Therapy Evaluation in the Netherlands (ATHENA) national observational HIV cohort was established to demonstrate the lifesaving effectiveness of triple combination antiretroviral therapy, including HIV-protease inhibitors, that had recently been made available for clinical use. Subsequently, the HIV Monitoring Foundation was established by the Dutch Ministry of Health, Welfare and Sport to continue ATHENA as an open cohort in order to continue the registration and monitoring of all HIV-positive people as an integral part of HIV care in all 26 HIV treatment centres in the Netherlands.
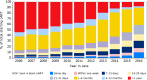
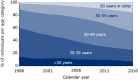
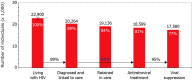
Participants: To date, a total of 25 036 participants have been enrolled in the cohort, with 263 600 person-years of follow-up. As of 1 January 2017, 19 035 HIV-1-positive participants were known to be in care: 18 824 adults (81% men and 19% women) and 211 children (47% boys and 53% girls). The remaining 6001 participants had either died (46%), were lost to care (29%) or had moved abroad (25%).
Findings to date: Today, with over 20 years of follow-up, the ATHENA cohort has provided extensive knowledge on HIV treatment, comorbidities and coinfections and created insight into the transmission dynamics of the HIV epidemic.
Future plans: ATHENA continues to enrol and monitor HIV positive people entering HIV care in the Netherlands. Future research will continue to provide tangible input into HIV care and prevention policies in the Netherlands and internationally.
Keywords: HIV & AIDS; epidemiology; infectious diseases; public health; virology.
© Author(s) (or their employer(s)) 2018. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures
References
-
- Stichting HIV Monitoring. ATHENA report 2001. Stichting HIV Monitoring, 2001. www.hiv-monitoring.nl.
-
- van Sighem AI, Boender TS, Wit F, et al. . Monitoring report 2017. Human immunodeficiency virus (hiv) infection in the Netherlands. Amsterdam. 2017. wwww.hiv-monitoring.nl.
-
- Hermanides HS, Gras L, Winkel CN, et al. . The efficacy of combination antiretroviral therapy in HIV type 1-infected patients treated in Curaçao compared with Antillean, Surinam, and Dutch HIV type 1-infected patients treated in The Netherlands. AIDS Res Hum Retroviruses 2011;27:605–12. 10.1089/aid.2010.0141 - DOI - PubMed
-
- Stichting HIV Monitoring. Jaarverslag 2016. Amsterdam: Stichting HIV Monitoring, 2017.
-
- Europese Unie. Regulation (EU) 2016/679 of the European parliament and of the council. Off J Eur Union 20162016;4–L119.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
