Growth inhibition and chemo-radiosensitization of esophageal squamous cell carcinoma by survivin-shRNA lentivirus transfection
- PMID: 30250546
- PMCID: PMC6144918
- DOI: 10.3892/ol.2018.9280
Growth inhibition and chemo-radiosensitization of esophageal squamous cell carcinoma by survivin-shRNA lentivirus transfection
Abstract
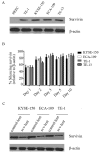
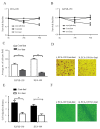
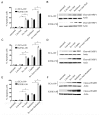
Esophageal cancer is one of the most common types of cancer worldwide, and it has a poor prognosis. Chemo-radiotherapy resistance and cancer relapse are among the most difficult issues in its treatment. Identifying the underlying molecular mechanisms is critical for developing novel therapies. Survivin has been previously suggested to be overexpressed in esophageal cancer cells. The present study identified that down-regulation of survivin sensitized esophageal cancer cells to chemo-radiotherapy. Consistent with previous studies, the present study indicated that survivin was overexpressed in 4 esophageal squamous carcinoma cell lines. Short hairpin RNA delivered by lentivirus successfully knocked down survivin in these cancer cell lines. Consequently, down-regulation of survivin impaired their colony-forming, migratory and invasive capabilities, while the overexpression of survivin in normal human esophagus epithelial cells improved their resistance to cisplatin, paclitaxel and radiation. Survivin knockdown induced apoptosis in esophageal cancer KYSE-150 and ECA-109 cell lines when exposed to the aforementioned chemo-radiotherapy treatments. These results indicate that survivin expression sustains growth in esophageal cancer cells, and confers resistance to chemo-radiotherapy. Targeted survivin ablation may be a promising strategy against esophageal tumor relapse and chemo-radioresistance.
Keywords: chemo-radiosensitization; esophageal cancer; lentivirus; short hairpin RNA; survivin.
Figures

Similar articles
-
Growth inhibition and chemo-radiosensitization of head and neck squamous cell carcinoma (HNSCC) by survivin-siRNA lentivirus.Radiother Oncol. 2016 Feb;118(2):359-68. doi: 10.1016/j.radonc.2015.12.007. Epub 2015 Dec 30. Radiother Oncol. 2016. PMID: 26747757
-
Overexpression of miR-214-3p in esophageal squamous cancer cells enhances sensitivity to cisplatin by targeting survivin directly and indirectly through CUG-BP1.Oncogene. 2016 Apr 21;35(16):2087-97. doi: 10.1038/onc.2015.271. Epub 2015 Aug 3. Oncogene. 2016. PMID: 26234674 Free PMC article.
-
Downregulation of Survivin by shRNA Inhibits Invasion and Enhances the Radiosensitivity of Laryngeal Squamous Cell Carcinoma.Cell Biochem Biophys. 2015 May;72(1):251-7. doi: 10.1007/s12013-014-0445-z. Cell Biochem Biophys. 2015. PMID: 25701406
-
Survivin, a molecular target for therapeutic interventions in squamous cell carcinoma.Cell Mol Biol Lett. 2017 Apr 5;22:8. doi: 10.1186/s11658-017-0038-0. eCollection 2017. Cell Mol Biol Lett. 2017. PMID: 28536639 Free PMC article. Review.
-
Genetic determinants for chemo- and radiotherapy resistance in bladder cancer.Transl Androl Urol. 2017 Dec;6(6):1081-1089. doi: 10.21037/tau.2017.08.19. Transl Androl Urol. 2017. PMID: 29354495 Free PMC article. Review.
Cited by
-
Translocating shRNA: a novel approach to RNA interference with Newcastle disease virus as viral vector.J Gen Virol. 2025 Jul;106(7):002127. doi: 10.1099/jgv.0.002127. J Gen Virol. 2025. PMID: 40643574 Free PMC article.
-
Analysis of cancer-promoting genes related to chemotherapy resistance in esophageal squamous cell carcinoma.Ann Transl Med. 2022 Jan;10(2):92. doi: 10.21037/atm-21-7032. Ann Transl Med. 2022. PMID: 35282117 Free PMC article.
-
Prognostic Value of BIRC5 in Lung Adenocarcinoma Lacking EGFR, KRAS, and ALK Mutations by Integrated Bioinformatics Analysis.Dis Markers. 2019 Apr 9;2019:5451290. doi: 10.1155/2019/5451290. eCollection 2019. Dis Markers. 2019. PMID: 31093306 Free PMC article.
-
The mechanisms and reversal strategies of tumor radioresistance in esophageal squamous cell carcinoma.J Cancer Res Clin Oncol. 2021 May;147(5):1275-1286. doi: 10.1007/s00432-020-03493-3. Epub 2021 Mar 9. J Cancer Res Clin Oncol. 2021. PMID: 33687564 Free PMC article. Review.
-
SREBP1 silencing inhibits the proliferation and motility of human esophageal squamous carcinoma cells via the Wnt/β-catenin signaling pathway.Oncol Lett. 2020 Sep;20(3):2855-2869. doi: 10.3892/ol.2020.11853. Epub 2020 Jul 10. Oncol Lett. 2020. PMID: 32765792 Free PMC article.
References
-
- van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BP, Richel DJ, Nieuwenhuijzen GA, Hospers GA, Bonenkamp JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–2084. doi: 10.1056/NEJMoa1112088. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
