Drug discovery and development: Biomarkers of neurotoxicity and neurodegeneration
- PMID: 30253665
- PMCID: PMC6434454
- DOI: 10.1177/1535370218801309
Drug discovery and development: Biomarkers of neurotoxicity and neurodegeneration
Abstract
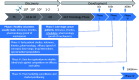
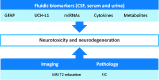
Attrition in drug discovery and development remains a major challenge. Safety/toxicity is the most prevalent reason for failure with cardiovascular and CNS toxicities predominating. Non-invasive biomarkers of neurotoxicity would provide significant advantage by allowing earlier prediction of likely neurotoxicity in preclinical studies as well as facilitating clinical trials of new therapies for neurodegenerative conditions such as Parkinson's disease (PD) and multiple sclerosis (MS).
Keywords: Biomarkers; central nervous system; drug development; drug safety; neurodegeneration; toxicology.
Figures


References
-
- Brennan R. Target safety assessment: strategies and resources In: Gautier JC. (ed) Drug safety evaluation. Methods in molecular biology. Vol.1641. New York, NY: Humana Press, 2017 - PubMed
-
- Roberts RA, Kavanagh S, Mellor H, Pollard C, Robinson S, Platz SJ. Reducing attrition in drug development: smart loading pre-clinical safety assessment. Drug Discov Today 2014; 19:341–7 - PubMed
-
- www.ICH.org (accessed 5 September 2018)
-
- Sanguinetti MC, Tristani-Firouzi M. hERG potassium channels and cardiac arrhythmia. Nature 2006; 440:463–9 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

