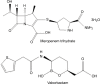
Overview of meropenem-vaborbactam and newer antimicrobial agents for the treatment of carbapenem-resistant Enterobacteriaceae
- PMID: 30254477
- PMCID: PMC6140735
- DOI: 10.2147/IDR.S150447
Overview of meropenem-vaborbactam and newer antimicrobial agents for the treatment of carbapenem-resistant Enterobacteriaceae
Abstract
There has been a worldwide increase in infections caused by drug-resistant Gram-negative pathogens, including carbapenem-resistant Enterobacteriaceae (CRE). Meropenem-vaborbactam, a carbapenem antibiotic and novel boronic acid-based beta-lactamase inhibitor, is a fixed-dose combination product with potent in vitro activity against Enterobacteriaceae that are Klebsiella pneumoniae carbapenemase producers. Meropenem-vaborbactam has been studied in two Phase III trials, Targeting Antibiotic Non-susceptible Gram-negative Organisms (TANGO)-I and TANGO-II. TANGO-I was a multicenter, international Phase III, randomized, double-blind, double-dummy, active-control trial to evaluate the efficacy and safety of meropenem-vaborbactam for the treatment of complicated urinary tract infection, including acute pyelonephritis. Among patients with complicated urinary tract infection and growth of a baseline pathogen, meropenem-vaborbactam was determined to be superior to piperacillin-tazobactam based on the composite outcome of symptom improvement or resolution and microbial eradication at the end of intravenous therapy. TANGO-II was a multicenter, international, Phase III, randomized, prospective, open-label, comparative trial to evaluate the efficacy and safety of meropenem-vaborbactam vs best available therapy for CRE infections. Treatment with meropenem-vaborbactam resulted in higher rates of clinical cure at the end of therapy (64.3%vs 33.3%, P=0.04). Additionally, 28-day all-cause mortality was 17.9% in the meropenem-vaborbactam group compared to 33.3% in the best available therapy group, a relative risk reduction of 46.5% (P=0.03). In addition to meropenem-vaborbactam, three other agents with activity against CRE are in late-stage development: imipenem-relebactam, plazomicin, and cefiderocol. The data from Phase II and III studies will help to further define the role of these agents. Overall, the recent approval of meropenem-vaborbactam and the active pipeline for other agents with broad Gram-negative activity are encouraging developments on the CRE therapeutic front.
Keywords: CRE; KPC; UTI; carbapenemase; meropenem; vaborbactam.
Conflict of interest statement
Disclosures KS Kaye, LA Petty, and JM Pogue are funded by the National Institute of Allergy and Infectious Diseases (DMID Protocol Number: 10-0065). KS Kaye and JM Pogue are funded by the National Institute of Allergy and Infectious Diseases (R01-AI119446-01). JM Pogue is a paid consultant, has received research support, and/or has served on the Speaker’s Bureau for The Medicines Company, Melinta, Merck, Allergand, Achaogen, Tetraphase, Shionogi, and Zavante. KS Kaye is a paid consultant and advisor for Melinta Therapeutics, The Medicines Company and Allergan. The authors have no other conflicts of interest in this work.
Figures
Similar articles
-
Imipenem-Relebactam and Meropenem-Vaborbactam: Two Novel Carbapenem-β-Lactamase Inhibitor Combinations.Drugs. 2018 Jan;78(1):65-98. doi: 10.1007/s40265-017-0851-9. Drugs. 2018. PMID: 29230684 Review.
-
An Appraisal of the Pharmacokinetic and Pharmacodynamic Properties of Meropenem-Vaborbactam.Infect Dis Ther. 2020 Dec;9(4):769-784. doi: 10.1007/s40121-020-00344-z. Epub 2020 Oct 6. Infect Dis Ther. 2020. PMID: 33025557 Free PMC article. Review.
-
Effect of Meropenem-Vaborbactam vs Piperacillin-Tazobactam on Clinical Cure or Improvement and Microbial Eradication in Complicated Urinary Tract Infection: The TANGO I Randomized Clinical Trial.JAMA. 2018 Feb 27;319(8):788-799. doi: 10.1001/jama.2018.0438. JAMA. 2018. PMID: 29486041 Free PMC article. Clinical Trial.
-
Meropenem/Vaborbactam: A Review in Complicated Urinary Tract Infections.Drugs. 2018 Aug;78(12):1259-1270. doi: 10.1007/s40265-018-0966-7. Drugs. 2018. PMID: 30128699 Free PMC article. Review.
-
An Update on Existing and Emerging Data for Meropenem-Vaborbactam.Clin Ther. 2020 Apr;42(4):692-702. doi: 10.1016/j.clinthera.2020.01.023. Epub 2020 Mar 5. Clin Ther. 2020. PMID: 32147146 Review.
Cited by
-
Biochemical exploration of β-lactamase inhibitors.Front Genet. 2023 Jan 17;13:1060736. doi: 10.3389/fgene.2022.1060736. eCollection 2022. Front Genet. 2023. PMID: 36733944 Free PMC article. Review.
-
Nosocomial Pneumonia in the Era of Multidrug-Resistance: Updates in Diagnosis and Management.Microorganisms. 2021 Mar 5;9(3):534. doi: 10.3390/microorganisms9030534. Microorganisms. 2021. PMID: 33807623 Free PMC article. Review.
-
In-vitro safety assessment of meropenem on human retinal pigment epithelium (RPE).Heliyon. 2024 Jul 1;10(14):e33916. doi: 10.1016/j.heliyon.2024.e33916. eCollection 2024 Jul 30. Heliyon. 2024. PMID: 39092242 Free PMC article.
-
Delafloxacin, Finafloxacin, and Zabofloxacin: Novel Fluoroquinolones in the Antibiotic Pipeline.Antibiotics (Basel). 2021 Dec 8;10(12):1506. doi: 10.3390/antibiotics10121506. Antibiotics (Basel). 2021. PMID: 34943718 Free PMC article. Review.
-
Meta-analysis of Clinical Outcomes Using Ceftazidime/Avibactam, Ceftolozane/Tazobactam, and Meropenem/Vaborbactam for the Treatment of Multidrug-Resistant Gram-Negative Infections.Open Forum Infect Dis. 2021 Jan 5;8(2):ofaa651. doi: 10.1093/ofid/ofaa651. eCollection 2021 Feb. Open Forum Infect Dis. 2021. PMID: 33598503 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases