The mechanochemical synthesis of quinazolin-4(3 H)-ones by controlling the reactivity of IBX
- PMID: 30254705
- PMCID: PMC6142747
- DOI: 10.3762/bjoc.14.216
The mechanochemical synthesis of quinazolin-4(3 H)-ones by controlling the reactivity of IBX
Abstract
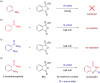
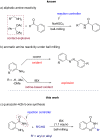
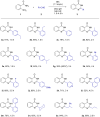
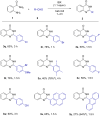
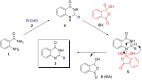
Performing any synthesis using several arylamines and hypervalent iodine(V) reagents by direct mixing is unrealistic because of the high exothermic reaction or explosion. Herein we demonstrate, when anilines were substituted with an amide group at the ortho-position, successful chemical reactions could be performed due to intramolecular control. At maximum contact of the reacting substances, i.e., under solvent-free mechanochemical conditions, 2-aminobenzamides, aryl-, alkylaldehydes and the iodine(V) reagent o-iodoxybenzoic acid (IBX) led to substituted quinazolin-4(3H)-one derivatives in fair yields.
Keywords: IBX; ball-mill; contact explosive; mechanochemical synthesis; quinazolin-4(3H)-one.
Figures

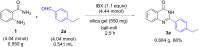
Similar articles
-
DDQ in mechanochemical C-N coupling reactions.Beilstein J Org Chem. 2022 Jun 1;18:639-646. doi: 10.3762/bjoc.18.64. eCollection 2022. Beilstein J Org Chem. 2022. PMID: 35706992 Free PMC article.
-
IBX-mediated synthesis of indazolone via oxidative N-N bond formation and unexpected formation of quinazolin-4-one: in situ generation of formaldehyde from dimethoxyethane.Arch Pharm Res. 2016 Mar;39(3):302-9. doi: 10.1007/s12272-016-0706-z. Epub 2016 Jan 16. Arch Pharm Res. 2016. PMID: 26780246
-
Quinazolin-4(3H)-ones and 5,6-Dihydropyrimidin-4(3H)-ones from β-Aminoamides and Orthoesters.Molecules. 2018 Nov 9;23(11):2925. doi: 10.3390/molecules23112925. Molecules. 2018. PMID: 30423947 Free PMC article.
-
2-Iodoxybenzoic acid organosulfonates: preparation, X-ray structure and reactivity of new, powerful hypervalent iodine(V) oxidants.Chem Commun (Camb). 2013 Dec 14;49(96):11269-71. doi: 10.1039/c3cc47090c. Epub 2013 Oct 24. Chem Commun (Camb). 2013. PMID: 24153437
-
IBX-Mediated Organic Transformations in Heterocyclic Chemistry-A Decade Update.Front Chem. 2022 Feb 28;10:841751. doi: 10.3389/fchem.2022.841751. eCollection 2022. Front Chem. 2022. PMID: 35295969 Free PMC article. Review.
Cited by
-
Iodoxybenzoic Acid (IBX) in Organic Synthesis: A Septennial Review.Curr Org Synth. 2024;21(5):607-664. doi: 10.2174/0115701794263252230924074035. Curr Org Synth. 2024. PMID: 37861006
-
DDQ in mechanochemical C-N coupling reactions.Beilstein J Org Chem. 2022 Jun 1;18:639-646. doi: 10.3762/bjoc.18.64. eCollection 2022. Beilstein J Org Chem. 2022. PMID: 35706992 Free PMC article.
-
Synthesis, in silico and in vitro antimicrobial efficacy of substituted arylidene-based quinazolin-4(3H)-one motifs.Front Chem. 2023 Sep 25;11:1264824. doi: 10.3389/fchem.2023.1264824. eCollection 2023. Front Chem. 2023. PMID: 37818483 Free PMC article.
-
Unveiling the Untapped Potential of Bertagnini's Salts in Microwave-Assisted Synthesis of Quinazolinones.Molecules. 2024 Apr 26;29(9):1986. doi: 10.3390/molecules29091986. Molecules. 2024. PMID: 38731478 Free PMC article.
References
-
- Tornieporth-Oetting I, Klapötke T. Angew Chem, Int Ed Engl. 1990;29:677–679. doi: 10.1002/anie.199006771. - DOI
-
- Dauban P, Dodd R H. Synlett. 2003:1571–1586. doi: 10.1055/s-2003-41010. - DOI
-
- Zhdankin V V. Hypervalent Iodine Chemistry. Chichester, UK: John Wiley & Sons Ltd; 2013. Preparation, Structure and Properties of Polyvalent Iodine Compounds; pp. 21–143.
LinkOut - more resources
Full Text Sources
Other Literature Sources
