Distinct Neuronal Projections From the Hypothalamic Ventromedial Nucleus Mediate Glycemic and Behavioral Effects
- PMID: 30257978
- PMCID: PMC6245222
- DOI: 10.2337/db18-0380
Distinct Neuronal Projections From the Hypothalamic Ventromedial Nucleus Mediate Glycemic and Behavioral Effects
Abstract
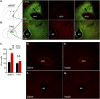
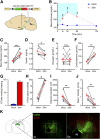
The hypothalamic ventromedial nucleus (VMN) is implicated both in autonomic control of blood glucose and in behaviors including fear and aggression, but whether these divergent effects involve the same or distinct neuronal subsets and their projections is unknown. To address this question, we used an optogenetic approach to selectively activate the subset of VMN neurons that express neuronal nitric oxide synthase 1 (VMNNOS1 neurons) implicated in glucose counterregulation. We found that photoactivation of these neurons elicits 1) robust hyperglycemia achieved by activation of counterregulatory responses usually reserved for the physiological response to hypoglycemia and 2) defensive immobility behavior. Moreover, we show that the glucagon, but not corticosterone, response to insulin-induced hypoglycemia is blunted by photoinhibition of the same neurons. To investigate the neurocircuitry by which VMNNOS1 neurons mediate these effects, and to determine whether these diverse effects are dissociable from one another, we activated downstream VMNNOS1 projections in either the anterior bed nucleus of the stria terminalis (aBNST) or the periaqueductal gray (PAG). Whereas glycemic responses are fully recapitulated by activation of VMNNOS1 projections to the aBNST, freezing immobility occurred only upon activation of VMNNOS1 terminals in the PAG. These findings support previous evidence of a VMN→aBNST neurocircuit involved in glucose counterregulation and demonstrate that activation of VMNNOS1 neuronal projections supplying the PAG robustly elicits defensive behaviors.
© 2018 by the American Diabetes Association.
Figures




References
-
- Ritter RC, Slusser PG, Stone S. Glucoreceptors controlling feeding and blood glucose: location in the hindbrain. Science 1981;213:451–452 - PubMed
-
- Saberi M, Bohland M, Donovan CM. The locus for hypoglycemic detection shifts with the rate of fall in glycemia: the role of portal-superior mesenteric vein glucose sensing. Diabetes 2008;57:1380–1386 - PubMed
-
- Beall C, Ashford ML, McCrimmon RJ. The physiology and pathophysiology of the neural control of the counterregulatory response. Am J Physiol Regul Integr Comp Physiol 2012;302:R215–R223 - PubMed
-
- Cryer PE. Mechanisms of hypoglycemia-associated autonomic failure in diabetes. N Engl J Med 2013;369:362–372 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

