Parahydrogen-Induced Polarization Study of the Silica-Supported Vanadium Oxo Organometallic Catalyst
- PMID: 30258526
- PMCID: PMC6150668
- DOI: 10.1021/acs.jpcc.7b12069
Parahydrogen-Induced Polarization Study of the Silica-Supported Vanadium Oxo Organometallic Catalyst
Abstract
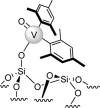
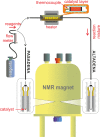
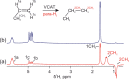
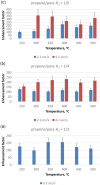
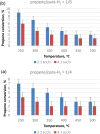
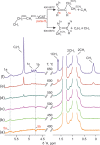
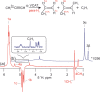
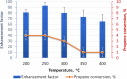
Parahydrogen can be used in catalytic hydrogenations to achieve substantial enhancement of NMR signals of the reaction products and in some cases of the reaction reagents as well. The corresponding nuclear spin hyperpolarization technique, known as parahydrogen-induced polarization (PHIP), has been applied to boost the sensitivity of NMR spectroscopy and magnetic resonance imaging by several orders of magnitude. The catalyst properties are of paramount importance for PHIP because the addition of parahydrogen to a substrate must be pairwise. This requirement significantly narrows down the range of the applicable catalysts. Herein, we study an efficient silica-supported vanadium oxo organometallic complex (VCAT) in hydrogenation and dehydrogenation reactions in terms of efficient PHIP production. This is the first example of group 5 catalyst used to produce PHIP. Hydrogenations of propene and propyne with parahydrogen over VCAT demonstrated production of hyperpolarized propane and propene, respectively. The achieved NMR signal enhancements were 200-300-fold in the case of propane and 1300-fold in the case of propene. Propane dehydrogenation in the presence of parahydrogen produced no hyperpolarized propane, but instead the hyperpolarized side-product 1-butene was detected. Test experiments of other group 5 (Ta) and group 4 (Zr) catalysts showed a much lower efficiency in PHIP as compared to that of VCAT. The results prove the general conclusion that vanadium-based catalysts and other group 4 and group 5 catalysts can be used to produce PHIP. The hydrogenation/dehydrogenation processes, however, are accompanied by side reactions leading, for example, to C4, C2, and C1 side products. Some of the side products like 1-butene and 2-butene were shown to appear hyperpolarized, demonstrating that the reaction mechanism includes pairwise parahydrogen addition in these cases as well.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
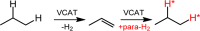
Similar articles
-
Heterogeneous addition of H2 to double and triple bonds over supported Pd catalysts: a parahydrogen-induced polarization technique study.Phys Chem Chem Phys. 2012 Aug 21;14(31):11008-14. doi: 10.1039/c2cp40690j. Epub 2012 Jul 5. Phys Chem Chem Phys. 2012. PMID: 22763714
-
Efficient Batch-Mode Parahydrogen-Induced Polarization of Propane.Chemphyschem. 2016 Nov 4;17(21):3395-3398. doi: 10.1002/cphc.201600564. Epub 2016 Sep 5. Chemphyschem. 2016. PMID: 27459542 Free PMC article.
-
Parahydrogen-induced polarization in heterogeneous catalytic processes.Top Curr Chem. 2013;338:123-80. doi: 10.1007/128_2012_371. Top Curr Chem. 2013. PMID: 23097028
-
Heterogeneous Catalysis and Parahydrogen-Induced Polarization.Chemphyschem. 2021 Jul 16;22(14):1421-1440. doi: 10.1002/cphc.202100153. Epub 2021 Jun 26. Chemphyschem. 2021. PMID: 33969590 Review.
-
Unconventional Parahydrogen-Induced Hyperpolarization Effects in Chemistry and Catalysis: From Photoreactions to Enzymes.ACS Catal. 2025 Apr 4;15(8):6386-6409. doi: 10.1021/acscatal.4c07870. eCollection 2025 Apr 18. ACS Catal. 2025. PMID: 40270879 Free PMC article. Review.
Cited by
-
Tailoring rhodium-based metal-organic layers for parahydrogen-induced polarization: achieving 20% polarization of 1H in liquid phase.Natl Sci Rev. 2024 Nov 13;12(1):nwae406. doi: 10.1093/nsr/nwae406. eCollection 2025 Jan. Natl Sci Rev. 2024. PMID: 39764503 Free PMC article.
-
Mechanistic Insights into Molecular Crystalline Organometallic Heterogeneous Catalysis through Parahydrogen-Based Nuclear Magnetic Resonance Studies.J Am Chem Soc. 2023 Feb 1;145(4):2619-2629. doi: 10.1021/jacs.2c12642. Epub 2023 Jan 23. J Am Chem Soc. 2023. PMID: 36688560 Free PMC article.
-
Parahydrogen-induced polarization with a metal-free P-P biradicaloid.Phys Chem Chem Phys. 2019 Mar 13;21(11):5890-5893. doi: 10.1039/c8cp07625a. Phys Chem Chem Phys. 2019. PMID: 30694276 Free PMC article.
-
Contrasting Metallic (Rh0) and Carbidic (2D-Mo2C MXene) Surfaces in Olefin Hydrogenation Provides Insights on the Origin of the Pairwise Hydrogen Addition.ACS Catal. 2024 Aug 6;14(16):12500-12511. doi: 10.1021/acscatal.4c02534. eCollection 2024 Aug 16. ACS Catal. 2024. PMID: 39169907 Free PMC article.
-
Advancing homogeneous catalysis for parahydrogen-derived hyperpolarisation and its NMR applications.Chem Sci. 2022 Mar 22;13(17):4670-4696. doi: 10.1039/d2sc00737a. eCollection 2022 May 4. Chem Sci. 2022. PMID: 35655870 Free PMC article. Review.
References
-
- Bowers C. R.; Weitekamp D. P. Parahydrogen and Synthesis Allow Dramatically Enhanced Nuclear Alignment. J. Am. Chem. Soc. 1987, 109, 5541–5542. 10.1021/ja00252a049. - DOI
-
- Natterer J.; Bargon J. Parahydrogen Induced Polarization. Prog. Nucl. Magn. Reson. Spectrosc. 1997, 31, 293–315. 10.1016/s0079-6565(97)00007-1. - DOI
-
- Kovtunov K. V.; Zhivonitko V. V.; Skovpin I. V.; Barskiy D. A.; Koptyug I. V.. Hyperpolarization Methods in NMR Spectroscopy; Kuhn L. T., Ed.; Topics in Current Chemistry; Springer: Berlin, Heidelberg, 2013; Vol. 338, pp 123–180. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
