Real-time examination of cAMP activity at relaxin family peptide receptors using a BRET-based biosensor
- PMID: 30263124
- PMCID: PMC6153321
- DOI: 10.1002/prp2.432
Real-time examination of cAMP activity at relaxin family peptide receptors using a BRET-based biosensor
Abstract
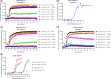
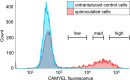
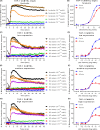
Relaxin family peptide (RXFPs) 1-4 receptors modulate the activity of cyclic adenosine monophosphate (cAMP) to produce a range of physiological functions. RXFP1 and RXFP2 increase cAMP via Gαs, whereas RXFP3 and RXFP4 inhibit cAMP via Gαi/o. RXFP1 also shows a delayed increase in cAMP downstream of Gαi3. In this study we have assessed whether the bioluminescence resonance energy transfer (BRET)-based biosensor CAMYEL (cAMP sensor using YFP-Epac-Rluc), which allows real-time measurement of cAMP activity in live cells, will aid in understanding ligand- and cell-specific RXFP signaling. CAMYEL detected concentration-dependent changes in cAMP activity at RXFP1-4 in recombinant cell lines, using a variety of ligands with potencies comparable to those seen in conventional cAMP assays. We used RXFP2 and RXFP3 antagonists to demonstrate that CAMYEL detects dynamic changes in cAMP by reversing cAMP activation or inhibition respectively, with real-time addition of antagonist after agonist stimulation. To demonstrate the utility of CAMYEL to detect cAMP activation in native cells expressing low levels of RXFP receptor, we cloned CAMYEL into a lentiviral vector and transduced THP-1 cells, which express low levels of RXFP1. THP-1 CAMYEL cells demonstrated robust cAMP activation in response to relaxin. However, the CAMYEL assay was unable to detect the Gαi3-mediated phase of RXFP1 cAMP activation in PTX-treated THP-1 cells or HEK293A cells with knockout of Gαs. Our data demonstrate that cytoplasmically-expressed CAMYEL efficiently detects real-time cAMP activation by Gαs or inhibition by Gαi/o but may not detect cAMP generated in specific intracellular compartments such as that generated by Gαi3 upon RXFP1 activation.
Keywords: CAMYEL; GPCR; RXFP; cAMP; relaxin; signalling.
Figures


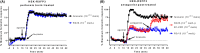



References
-
- Bathgate RA, Kocan M, Scott DJ, et al. The relaxin receptor as a therapeutic target – perspectives from evolution and drug targeting. Pharmacol Ther. 2018;187:114‐132. - PubMed
-
- Bathgate RAD, Halls ML, van der Westhuizen ET, Callander GE, Kocan M, Summers RJ. Relaxin family peptides and their receptors. Physiol Rev. 2013;93:405‐480. - PubMed
-
- Bathgate RA, Ivell R, Sanborn BM, Sherwood OD, Summers RJ. International Union of Pharmacology LVII: recommendations for the nomenclature of receptors for relaxin family peptides. Pharmacol Rev. 2006;58:7‐31. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

