Ibuprofen for the treatment of patent ductus arteriosus in preterm or low birth weight (or both) infants
- PMID: 30264852
- PMCID: PMC6513618
- DOI: 10.1002/14651858.CD003481.pub7
Ibuprofen for the treatment of patent ductus arteriosus in preterm or low birth weight (or both) infants
Update in
-
Ibuprofen for the treatment of patent ductus arteriosus in preterm or low birth weight (or both) infants.Cochrane Database Syst Rev. 2020 Feb 11;2(2):CD003481. doi: 10.1002/14651858.CD003481.pub8. Cochrane Database Syst Rev. 2020. PMID: 32045960 Free PMC article.
Abstract
Background: Indomethacin is used as standard therapy to close a patent ductus arteriosus (PDA) but is associated with reduced blood flow to several organs. Ibuprofen, another cyclo-oxygenase inhibitor, may be as effective as indomethacin with fewer adverse effects.
Objectives: To determine the effectiveness and safety of ibuprofen compared with indomethacin, other cyclo-oxygenase inhibitor(s), placebo, or no intervention for closing a patent ductus arteriosus in preterm, low-birth-weight, or preterm and low-birth-weight infants.
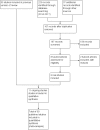
Search methods: We used the standard search strategy of Cochrane Neonatal to search the Cochrane Central Register of Controlled Trials (CENTRAL 2017, Issue 10), MEDLINE via PubMed (1966 to 30 November 2017), Embase (1980 to 30 November 2017), and CINAHL (1982 to 30 November 2017). We searched clinical trials databases, conference proceedings, and the reference lists of retrieved articles for randomised controlled trials and quasi-randomised trials.
Selection criteria: Randomised or quasi-randomised controlled trials of ibuprofen for the treatment of a PDA in preterm, low birth weight, or both preterm and low-birth-weight newborn infants.
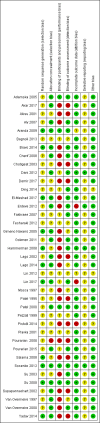
Data collection and analysis: Data collection and analysis conformed to the methods of the Cochrane Neonatal Review Group. We used the GRADE approach to assess the quality of evidence.
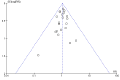
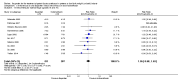
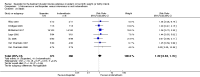
Main results: We included 39 studies enrolling 2843 infants.Ibuprofen (IV) versus placebo: IV Ibuprofen (3 doses) reduced the failure to close a PDA compared with placebo (typical relative risk (RR); 0.62 (95% CI 0.44 to 0.86); typical risk difference (RD); -0.18 (95% CI -0.30 to -0.06); NNTB 6 (95% CI 3 to 17); I2 = 65% for RR and I2 = 0% for RD; 2 studies, 206 infants; moderate-quality the evidence). One study reported decreased failure to close a PDA after single or three doses of oral ibuprofen compared with placebo (64 infants; RR 0.26, 95% CI 0.11 to 0.62; RD -0.44, 95% CI -0.65 to -0.23; NNTB 2, 95% CI 2 to 4; I2 test not applicable).Ibuprofen (IV or oral) compared with indomethacin (IV or oral): Twenty-four studies (1590 infants) comparing ibuprofen (IV or oral) with indomethacin (IV or oral) found no significant differences in failure rates for PDA closure (typical RR 1.07, 95% CI 0.92 to 1.24; typical RD 0.02, 95% CI -0.02 to 0.06; I2 = 0% for both RR and RD; moderate-quality evidence). A reduction in NEC (necrotising enterocolitis) was noted in the ibuprofen (IV or oral) group (18 studies, 1292 infants; typical RR 0.68, 95% CI 0.49 to 0.94; typical RD -0.04, 95% CI -0.07 to -0.01; NNTB 25, 95% CI 14 to 100; I2 = 0% for both RR and RD; moderate-quality evidence). There was a statistically significant reduction in the proportion of infants with oliguria in the ibuprofen group (6 studies, 576 infants; typical RR 0.28, 95% CI 0.14 to 0.54; typical RD -0.09, 95% CI -0.14 to -0.05; NNTB 11, 95% CI 7 to 20; I2 = 24% for RR and I2 = 69% for RD; moderate-quality evidence). The serum/plasma creatinine levels 72 hours after initiation of treatment were statistically significantly lower in the ibuprofen group (11 studies, 918 infants; MD -8.12 µmol/L, 95% CI -10.81 to -5.43). For this comparison, there was high between-study heterogeneity (I2 = 83%) and low-quality evidence.Ibuprofen (oral) compared with indomethacin (IV or oral): Eight studies (272 infants) reported on failure rates for PDA closure in a subgroup of the above studies comparing oral ibuprofen with indomethacin (IV or oral). There was no significant difference between the groups (typical RR 0.96, 95% CI 0.73 to 1.27; typical RD -0.01, 95% CI -0.12 to 0.09; I2 = 0% for both RR and RD). The risk of NEC was reduced with oral ibuprofen compared with indomethacin (IV or oral) (7 studies, 249 infants; typical RR 0.41, 95% CI 0.23 to 0.73; typical RD -0.13, 95% CI -0.22 to -0.05; NNTB 8, 95% CI 5 to 20; I2 = 0% for both RR and RD). There was low-quality evidence for these two outcomes. There was a decreased risk of failure to close a PDA with oral ibuprofen compared with IV ibuprofen (5 studies, 406 infants; typical RR 0.38, 95% CI 0.26 to 0.56; typical RD -0.22, 95% CI -0.31 to -0.14; NNTB 5, 95% CI 3 to 7; moderate-quality evidence). There was a decreased risk of failure to close a PDA with high-dose versus standard-dose of IV ibuprofen (3 studies 190 infants; typical RR 0.37, 95% CI 0.22 to 0.61; typical RD - 0.26, 95% CI -0.38 to -0.15; NNTB 4, 95% CI 3 to 7); I2 = 4% for RR and 0% for RD); moderate-quality evidence).Early versus expectant administration of IV ibuprofen, echocardiographically-guided IV ibuprofen treatment versus standard IV ibuprofen treatment, continuous infusion of ibuprofen versus intermittent boluses of ibuprofen, and rectal ibuprofen versus oral ibuprofen were studied in too few trials to allow for precise estimates of any clinical outcomes.
Authors' conclusions: Ibuprofen is as effective as indomethacin in closing a PDA. Ibuprofen reduces the risk of NEC and transient renal insufficiency. Therefore, of these two drugs, ibuprofen appears to be the drug of choice. The effectiveness of ibuprofen versus paracetamol is assessed in a separate review. Oro-gastric administration of ibuprofen appears as effective as IV administration. To make further recommendations, studies are needed to assess the effectiveness of high-dose versus standard-dose ibuprofen, early versus expectant administration of ibuprofen, echocardiographically-guided versus standard IV ibuprofen, and continuous infusion versus intermittent boluses of ibuprofen. Studies are lacking evaluating the effect of ibuprofen on longer-term outcomes in infants with PDA.
Conflict of interest statement
Arne Ohlsson ‐ none known.
Rajneesh Walia ‐ none known.
Sachin Shah ‐ none known.
Figures

































































































































































Update of
-
Ibuprofen for the treatment of patent ductus arteriosus in preterm or low birth weight (or both) infants.Cochrane Database Syst Rev. 2015 Feb 18;(2):CD003481. doi: 10.1002/14651858.CD003481.pub6. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2018 Sep 28;9:CD003481. doi: 10.1002/14651858.CD003481.pub7. PMID: 25692606 Updated.
References
References to studies included in this review
-
- Adamska E, Helwich E, Rutkowska M, Zacharska E, Piotrowska A. Comparison of the efficacy of ibuprofen and indomethacin in the treatment of patent ductus arteriosus in prematurely born infants [Porownanie ibuprofenu i indometacyny w leczeniu przetrwalego przewodu tetniczego u noworodkow urodzonych przedwczesnie]. Medycyna Wieku Rozwojowego 2005;9(3 Pt 1):335‐54. [PUBMED: 16547381] - PubMed
-
- Akar M, Yildirim TG, Sandal G, Bozdag S, Erdeve O, Altug N, et al. Does ibuprofen treatment in patent ductus arteriosus alter oxygen free radicals in premature infants?. Cardiology in the Young 2017;27(3):507–11. [PUBMED: 27319277] - PubMed
-
- Akisu M, Ozyurek AR, Dorak C, Parlar A, Kultursay N. Enteral ibuprofen versus indomethacin in the treatment of patent ductus arteriosus in preterm newborn infants [Premature bebeklerde patent duktus arteriozusun tedavisinde enteral ibuprofen ve indometazinin etkinligi ve guvenilirligi]. Cocuk Sagligi ve Hastaliklari Dergisi 2001;44(1):56‐60.
-
- Aly H, Lotfy W, Badrawi N, Ghawas M, Abdel‐Meguid IE, Hammad TA. Oral ibuprofen and ductus arteriosus in premature infants: a randomized pilot study. American Journal of Perinatology 2007;24(5):267‐70. [PUBMED: 17484080] - PubMed
- Lotfy W, Badrawi N, Ghawas M, Ehsan E, Aly H. Oral ibuprofen solution (O) is efficacious for the treatment of patent ductus arteriosus (PDA) in premature infants: a randomized controlled trial. Pediatric Academic Societies Annual Meeting; 2005 May 14‐17; Washington DC, United States. 2005. [PAS2005:1410]
-
- Aranda JV. Multicentre randomized double‐blind placebo controlled trial of ibuprofen L‐Lysine intravenous solution (IV Ibuprofen) in premature infants for the early treatment of patent ductus arteriosus (PDA). Pediatric Academic Societies Annual Meeting; 2005 May 14‐17; Washington DC, United States. 2005.
- Aranda JV, Clyman R, Cox B, Overmeire B, Wozniak P, Sosenko I, et al. A randomized, double‐blind, placebo‐controlled trial of intravenous ibuprofen L‐lysine for the early closure of non‐symptomatic patent ductus arteriosus within 72 hours of birth in extremely low‐birth‐weight infants. American Journal of Perinatology 2009;26(3):235‐45. [DOI: 10.1055/s-0028-1103515; PUBMED: 19067286] - DOI - PubMed
References to studies excluded from this review
-
- Alipour MR, Shamsi MM, Namayandeh SM, Pezeshkpour Z, Rezaeipour F, Sarebanhassanabadi M. The effects of oral ibuprofen on medicinal closure of patent ductus arteriosus in full‐term neonates in the second postnatal week. Iranian Journal of Pediatrics 2016;26(4):e5807. [DOI: 10.5812/ijp.5807; PUBMED: 27729962] - DOI - PMC - PubMed
-
- Desfrere L, Zohar S, Morville P, Brunhes A, Chevret S, Pons G, et al. Dose‐finding study of ibuprofen in patent ductus arteriosus using the continual reassessment method. Journal of Clinical Pharmacy and Therapeutics 2005;30(2):121‐32. [DOI: 10.1111/j.1365-2710.2005.00630.x; PUBMED: 15811164] - DOI - PubMed
-
- Kalani M, Shariat M, Khalesi N, Farahani Z, Ahmadi S. A comparison of early ibuprofen and indomethacin administration to prevent intraventricular hemorrhage among preterm infants. Acta Medica Iranica 2016;54(12):788‐92. [PUBMED: 28120591] - PubMed
References to ongoing studies
-
- ACTRN12616000195459. Early pharmacological treatment with supportive care versus supportive care alone in preterm infants with a patent ductus arteriosus. anzctr.org.au/ACTRN12616000195459.aspx (first received 08 February 2016).
-
- ChiCTR‐TRC‐14004559. Comparison of the dose effect of oral ibuprofen suspension for PDA treatment in premature infants [Developmental pharmacokinetics and pharmacodynamics of chiral ibuprofen associated with the CYP2C8/9 gene polymorphism]. chictr.org.cn/showproj.aspx?proj=5014 (first received 15 April 2014).
-
- EUCTR2016‐002974‐11‐ES. Clinical trial to evaluate the impact on the intestinal prognosis of 2 ibuprofen administration regimens for the treatment of patent ductus arteriosus, guided by echocardiography [Phase III, randomized, multicenter, double‐blind clinical trial to evaluate two echo‐guided administration regimens of ibuprofen in the treatment of patent ductus arteriosus: impact on intestinal prognosis]. clinicaltrialsregister.eu/ctr‐search/search?query=eudract_number:2016‐00... (first received 26 September 2016).
-
- IRCT201205029611N1. High dose oral ibuprofen in PDA closure in premature infants. en.irct.ir/trial/10178 (first received 25 August 2014).
-
- IRCT2015111024977N1. Comparison of oral Ibuprofen and intravenous indomethacin for the treatment of patent ductus arteriosus [Comparison of oral Ibuprofen and intravenous indomethacin for the treatment of patent ductus arteriosus in preterm infants]. en.irct.ir/trial/20973 (first received 16 December 2015).
Additional references
-
- Chemtob S, Behary K, Rex J, Varma DR, Aranda JV. Prostanoids determine the range of cerebral blood flow autoregulation of newborn piglets. Stroke 1990;21(5):777‐84. [PUBMED: 2339458] - PubMed
-
- Chemtob S, Beharry K, Barna T, Varma DR, Aranda JV. Differences in the effects in the newborn piglet of various nonsteroidal antiinflammatory drugs on cerebral blood flow but not on cerebrovascular prostaglandins. Pediatric Research 1991;30(1):106‐11. [DOI: 10.1203/00006450-199107000-00021; PUBMED: 1891274] - DOI - PubMed
References to other published versions of this review
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

