Generation of mouse-zebrafish hematopoietic tissue chimeric embryos for hematopoiesis and host-pathogen interaction studies
- PMID: 30266803
- PMCID: PMC6262816
- DOI: 10.1242/dmm.034876
Generation of mouse-zebrafish hematopoietic tissue chimeric embryos for hematopoiesis and host-pathogen interaction studies
Abstract
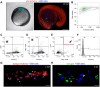
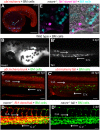
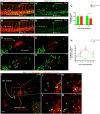
Xenografts of the hematopoietic system are extremely useful as disease models and for translational research. Zebrafish xenografts have been widely used to monitor blood cancer cell dissemination and homing due to the optical clarity of embryos and larvae, which allow unrestricted in vivo visualization of migratory events. Here, we have developed a xenotransplantation technique that transiently generates hundreds of hematopoietic tissue chimeric embryos by transplanting murine bone marrow cells into zebrafish blastulae. In contrast to previous methods, this procedure allows mammalian cell integration into the fish developmental hematopoietic program, which results in chimeric animals containing distinct phenotypes of murine blood cells in both circulation and the hematopoietic niche. Murine cells in chimeric animals express antigens related to (i) hematopoietic stem and progenitor cells, (ii) active cell proliferation and (iii) myeloid cell lineages. We verified the utility of this method by monitoring zebrafish chimeras during development using in vivo non-invasive imaging to show novel murine cell behaviors, such as homing to primitive and definitive hematopoietic tissues, dynamic hematopoietic cell and hematopoietic niche interactions, and response to bacterial infection. Overall, transplantation into the zebrafish blastula provides a useful method that simplifies the generation of numerous chimeric animals and expands the range of murine cell behaviors that can be studied in zebrafish chimeras. In addition, integration of murine cells into the host hematopoietic system during development suggests highly conserved molecular mechanisms of hematopoiesis between zebrafish and mammals.This article has an associated First Person interview with the first author of the paper.
Keywords: Cell migration; Hematopoiesis; Host-pathogen interactions; Live imaging; Xenotransplantation; Zebrafish.
© 2018. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

