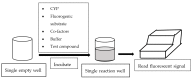
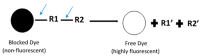Current High-Throughput Approaches of Screening Modulatory Effects of Xenobiotics on Cytochrome P450 (CYP) Enzymes
- PMID: 30274310
- PMCID: PMC6306765
- DOI: 10.3390/ht7040029
Current High-Throughput Approaches of Screening Modulatory Effects of Xenobiotics on Cytochrome P450 (CYP) Enzymes
Abstract
Cytochrome P450 (CYP) is a critical drug-metabolizing enzyme superfamily. Modulation of CYP enzyme activities has the potential to cause drug⁻drug/herb interactions. Drug⁻drug/herb interactions can lead to serious adverse drug reactions (ADRs) or drug failures. Therefore, there is a need to examine the modulatory effects of new drug entities or herbal preparations on a wide range of CYP isoforms. The classic method of quantifying CYP enzyme activities is based on high-performance liquid chromatography (HPLC), which is time- and reagent-consuming. In the past two decades, high-throughput screening methods including fluorescence-based, luminescence-based, and mass-spectrometry-based assays have been developed and widely applied to estimate CYP enzyme activities. In general, these methods are faster and use lower volume of reagents than HPLC. However, each high-throughput method has its own limitations. Investigators may make a selection of these methods based on the available equipment in the laboratory, budget, and enzyme sources supplied. Furthermore, the current high-throughput systems should look into developing a reliable automation mechanism to accomplish ultra-high-throughput screening in the near future.
Keywords: cytochrome P450; high-throughput screening; in vitro; inhibitory effects.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Fluorescence-Based High-Throughput Assays for Investigating Cytochrome P450 Enzyme-Mediated Drug-Drug Interactions.Drug Metab Dispos. 2023 Oct;51(10):1254-1272. doi: 10.1124/dmd.122.001068. Epub 2023 Jun 22. Drug Metab Dispos. 2023. PMID: 37349113 Review.
-
Reliable high-throughput method for inhibition assay of 8 cytochrome P450 isoforms using cocktail of probe substrates and stable isotope-labeled internal standards.Drug Metab Pharmacokinet. 2012;27(5):520-9. doi: 10.2133/dmpk.dmpk-12-rg-014. Epub 2012 Apr 10. Drug Metab Pharmacokinet. 2012. PMID: 22498647
-
Interactions of herbs with cytochrome P450.Drug Metab Rev. 2003 Feb;35(1):35-98. doi: 10.1081/dmr-120018248. Drug Metab Rev. 2003. PMID: 12635815 Review.
-
Cytochrome P450 Structure, Function and Clinical Significance: A Review.Curr Drug Targets. 2018;19(1):38-54. doi: 10.2174/1389450118666170125144557. Curr Drug Targets. 2018. PMID: 28124606 Review.
-
High-throughput screening of inhibitory effects of Bo-yang-hwan-o-tang on human cytochrome P450 isoforms in vitro using UPLC/MS/MS.Anal Sci. 2012;28(12):1197-201. doi: 10.2116/analsci.28.1197. Anal Sci. 2012. PMID: 23232241
Cited by
-
Design and application of synthetic 17B-HSD13 substrates reveals preserved catalytic activity of protective human variants.Nat Commun. 2025 Jan 2;16(1):297. doi: 10.1038/s41467-024-54487-5. Nat Commun. 2025. PMID: 39746932 Free PMC article.
-
Bielectrode Strategy for Determination of CYP2E1 Catalytic Activity: Electrodes with Bactosomes and Voltammetric Determination of 6-Hydroxychlorzoxazone.Biomedicines. 2024 Jan 11;12(1):152. doi: 10.3390/biomedicines12010152. Biomedicines. 2024. PMID: 38255257 Free PMC article.
-
Effect of Naoxintong Capsules on the Activities of CYP450 and Metabolism of Metoprolol Tartrate in Rats Evaluated by Probe Cocktail and Pharmacokinetic Methods.Evid Based Complement Alternat Med. 2019 Sep 23;2019:5242605. doi: 10.1155/2019/5242605. eCollection 2019. Evid Based Complement Alternat Med. 2019. PMID: 31662775 Free PMC article.
-
How Plant Polyhydroxy Flavonoids Can Hinder the Metabolism of Cytochrome 3A4.Biomedicines. 2025 Mar 7;13(3):655. doi: 10.3390/biomedicines13030655. Biomedicines. 2025. PMID: 40149631 Free PMC article.
-
Development and Characterization of pFluor50, a Fluorogenic-Based Kinetic Assay System for High-Throughput Inhibition Screening and Characterization of Time-Dependent Inhibition and Inhibition Type for Six Human CYPs.Molecules. 2025 May 2;30(9):2032. doi: 10.3390/molecules30092032. Molecules. 2025. PMID: 40363839 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous