A Kirchhoff-Nernst-Planck framework for modeling large scale extracellular electrodiffusion surrounding morphologically detailed neurons
- PMID: 30286073
- PMCID: PMC6191143
- DOI: 10.1371/journal.pcbi.1006510
A Kirchhoff-Nernst-Planck framework for modeling large scale extracellular electrodiffusion surrounding morphologically detailed neurons
Abstract
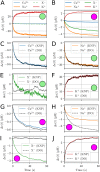
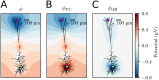
Many pathological conditions, such as seizures, stroke, and spreading depression, are associated with substantial changes in ion concentrations in the extracellular space (ECS) of the brain. An understanding of the mechanisms that govern ECS concentration dynamics may be a prerequisite for understanding such pathologies. To estimate the transport of ions due to electrodiffusive effects, one must keep track of both the ion concentrations and the electric potential simultaneously in the relevant regions of the brain. Although this is currently unfeasible experimentally, it is in principle achievable with computational models based on biophysical principles and constraints. Previous computational models of extracellular ion-concentration dynamics have required extensive computing power, and therefore have been limited to either phenomena on very small spatiotemporal scales (micrometers and milliseconds), or simplified and idealized 1-dimensional (1-D) transport processes on a larger scale. Here, we present the 3-D Kirchhoff-Nernst-Planck (KNP) framework, tailored to explore electrodiffusive effects on large spatiotemporal scales. By assuming electroneutrality, the KNP-framework circumvents charge-relaxation processes on the spatiotemporal scales of nanometers and nanoseconds, and makes it feasible to run simulations on the spatiotemporal scales of millimeters and seconds on a standard desktop computer. In the present work, we use the 3-D KNP framework to simulate the dynamics of ion concentrations and the electrical potential surrounding a morphologically detailed pyramidal cell. In addition to elucidating the single neuron contribution to electrodiffusive effects in the ECS, the simulation demonstrates the efficiency of the 3-D KNP framework. We envision that future applications of the framework to more complex and biologically realistic systems will be useful in exploring pathological conditions associated with large concentration variations in the ECS.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Effect of Ionic Diffusion on Extracellular Potentials in Neural Tissue.PLoS Comput Biol. 2016 Nov 7;12(11):e1005193. doi: 10.1371/journal.pcbi.1005193. eCollection 2016 Nov. PLoS Comput Biol. 2016. PMID: 27820827 Free PMC article.
-
An electrodiffusive neuron-extracellular-glia model for exploring the genesis of slow potentials in the brain.PLoS Comput Biol. 2021 Jul 16;17(7):e1008143. doi: 10.1371/journal.pcbi.1008143. eCollection 2021 Jul. PLoS Comput Biol. 2021. PMID: 34270543 Free PMC article.
-
Modeling extracellular space electrodiffusion during Leão's spreading depression.IEEE Trans Biomed Eng. 2004 Mar;51(3):450-8. doi: 10.1109/TBME.2003.821010. IEEE Trans Biomed Eng. 2004. PMID: 15000376
-
ECS Dynamism and Its Influence on Neuronal Excitability and Seizures.Neurochem Res. 2019 May;44(5):1020-1036. doi: 10.1007/s11064-019-02773-w. Epub 2019 Mar 16. Neurochem Res. 2019. PMID: 30879174 Review.
-
Contribution of dead-space microdomains to tortuosity of brain extracellular space.Neurochem Int. 2004 Sep;45(4):467-77. doi: 10.1016/j.neuint.2003.11.011. Neurochem Int. 2004. PMID: 15186912 Review.
Cited by
-
Conductance of porous media depends on external electric fields.Biophys J. 2021 Apr 20;120(8):1431-1442. doi: 10.1016/j.bpj.2021.02.012. Epub 2021 Feb 18. Biophys J. 2021. PMID: 33609495 Free PMC article.
-
Nano-scale solution of the Poisson-Nernst-Planck (PNP) equations in a fraction of two neighboring cells reveals the magnitude of intercellular electrochemical waves.PLoS Comput Biol. 2023 Feb 15;19(2):e1010895. doi: 10.1371/journal.pcbi.1010895. eCollection 2023 Feb. PLoS Comput Biol. 2023. PMID: 36791152 Free PMC article.
-
Investigate channel rectifications and neural dynamics by an electrodiffusive Gauss-Nernst-Planck approach.PLoS Comput Biol. 2025 Jun 30;21(6):e1012883. doi: 10.1371/journal.pcbi.1012883. eCollection 2025 Jun. PLoS Comput Biol. 2025. PMID: 40587520 Free PMC article.
-
An electrodiffusive, ion conserving Pinsky-Rinzel model with homeostatic mechanisms.PLoS Comput Biol. 2020 Apr 29;16(4):e1007661. doi: 10.1371/journal.pcbi.1007661. eCollection 2020 Apr. PLoS Comput Biol. 2020. PMID: 32348299 Free PMC article.
-
Using NEURON for Reaction-Diffusion Modeling of Extracellular Dynamics.Front Neuroinform. 2018 Jul 10;12:41. doi: 10.3389/fninf.2018.00041. eCollection 2018. Front Neuroinform. 2018. PMID: 30042670 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

