Conotoxin MVIIA improves cell viability and antioxidant system after spinal cord injury in rats
- PMID: 30286181
- PMCID: PMC6171875
- DOI: 10.1371/journal.pone.0204948
Conotoxin MVIIA improves cell viability and antioxidant system after spinal cord injury in rats
Abstract
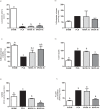
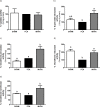
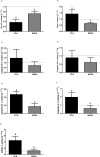
This study evaluates whether intrathecal MVIIA injection after spinal cord injury (SCI) elicits neuroprotective effects. The test rats were randomly distributed into six groups- sham, placebo, MVIIA 2.5 μM, MVIIA 5 μM, MVIIA 10 μM, and MVIIA 20 μM-and were administered the treatment four hours after SCI. After the optimal MVIIA dose (MVIIA 10 μM) was defined, the best time for application, one or four hours, was analyzed. Locomotor hind limb function and side effects were assessed. Forty-eight hours after the injury and immediately after euthanasia, spinal cord segments were removed from the test rats. Cell viability, reactive oxygen species, lipid peroxidation, and glutamate release were investigated. To examine the MVIIA mechanism of action, the gene expressions of pro-apoptotic (Bax, nNOS, and caspase-3, -8, -9, -12) and anti-apoptotic (Bcl-xl) factors in the spinal cord tissue samples were determined by real-time PCR, and the activities of antioxidant enzymes were also investigated. Application of intrathecal MVIIA 10 μM four hours after SCI prompted a neuroprotective effect: neuronal death decreased (22.46%), oxidative stress diminished, pro-apoptotic factors (Bax, nNOS, and caspase-3, -8) were expressed to a lesser extent, and mitochondrial viability as well as anti-apoptotic factor (Bcl-xl) expression increased. These results suggested that MVIIA provided neuroprotection through antioxidant effects. Indeed, superoxide dismutase (188.41%), and glutathione peroxidase (199.96%), reductase (193.86%), and transferase (175.93%) expressions increased. Therefore, intrathecal MVIIA (MVIIA 10 μM, 4 h) application has neuroprotective potential, and the possible mechanisms are related to antioxidant agent modulation and to intrinsic and extrinsic apoptotic pathways.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Neuroprotective effects of lycopene in spinal cord injury in rats via antioxidative and anti-apoptotic pathway.Neurosci Lett. 2017 Mar 6;642:107-112. doi: 10.1016/j.neulet.2017.02.004. Epub 2017 Feb 3. Neurosci Lett. 2017. PMID: 28163080
-
Hypericum perforatum Attenuates Spinal Cord Injury-Induced Oxidative Stress and Apoptosis in the Dorsal Root Ganglion of Rats: Involvement of TRPM2 and TRPV1 Channels.Mol Neurobiol. 2016 Aug;53(6):3540-3551. doi: 10.1007/s12035-015-9292-1. Epub 2015 Jun 23. Mol Neurobiol. 2016. PMID: 26099309
-
Epidermal growth factor regulates apoptosis and oxidative stress in a rat model of spinal cord injury.Injury. 2018 Jun;49(6):1038-1045. doi: 10.1016/j.injury.2018.03.021. Epub 2018 Mar 22. Injury. 2018. PMID: 29602490
-
Omega-3 fatty acids and neurological injury.Prostaglandins Leukot Essent Fatty Acids. 2007 Nov-Dec;77(5-6):295-300. doi: 10.1016/j.plefa.2007.10.021. Epub 2007 Nov 26. Prostaglandins Leukot Essent Fatty Acids. 2007. PMID: 18036801 Review.
-
The Importance of Natural Antioxidants in the Treatment of Spinal Cord Injury in Animal Models: An Overview.Oxid Med Cell Longev. 2019 Nov 12;2019:3642491. doi: 10.1155/2019/3642491. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 32676138 Free PMC article. Review.
Cited by
-
Omega-conotoxin MVIIA reduces neuropathic pain after spinal cord injury by inhibiting N-type voltage-dependent calcium channels on spinal dorsal horn.Front Neurosci. 2024 Feb 26;18:1366829. doi: 10.3389/fnins.2024.1366829. eCollection 2024. Front Neurosci. 2024. PMID: 38469570 Free PMC article.
-
Navigating neurological disorders: harnessing the power of natural compounds for innovative therapeutic breakthroughs.EXCLI J. 2024 Apr 23;23:534-569. doi: 10.17179/excli2024-7051. eCollection 2024. EXCLI J. 2024. PMID: 38741726 Free PMC article. Review.
-
Kisspeptin-10 Improves Testicular Redox Status but Does Not Alter the Unfolded Protein Response (UPR) That Is Downregulated by Hypothyroidism in a Rat Model.Int J Mol Sci. 2024 Jan 26;25(3):1514. doi: 10.3390/ijms25031514. Int J Mol Sci. 2024. PMID: 38338793 Free PMC article.
-
Peptide and Peptidomimetic Inhibitors Targeting the Interaction of Collapsin Response Mediator Protein 2 with the N-Type Calcium Channel for Pain Relief.ACS Pharmacol Transl Sci. 2024 Jun 6;7(7):1916-1936. doi: 10.1021/acsptsci.4c00181. eCollection 2024 Jul 12. ACS Pharmacol Transl Sci. 2024. PMID: 39022365 Free PMC article. Review.
-
Neurotoxins Acting at Synaptic Sites: A Brief Review on Mechanisms and Clinical Applications.Toxins (Basel). 2022 Dec 27;15(1):18. doi: 10.3390/toxins15010018. Toxins (Basel). 2022. PMID: 36668838 Free PMC article. Review.
References
-
- Oyinbo CA. Secondary injury mechanisms in traumatic spinal cord injury: a nugget of this multiply cascade. Acta Neurobiol Exp. 2011; 71:281–299. . - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

