Functionalized Gold Nanoparticles as Biosensors for Monitoring Cellular Uptake and Localization in Normal and Tumor Prostatic Cells
- PMID: 30287746
- PMCID: PMC6316160
- DOI: 10.3390/bios8040087
Functionalized Gold Nanoparticles as Biosensors for Monitoring Cellular Uptake and Localization in Normal and Tumor Prostatic Cells
Abstract
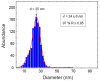
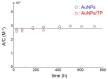
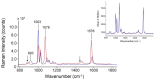
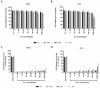
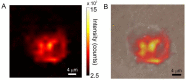
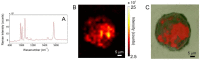
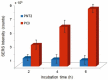
In the present contribution the fabrication and characterization of functionalized gold nanospheres of uniform shape and controlled size is reported. These nano-objects are intended to be used as Surface Enhanced Raman Spectroscopy (SERS) sensors for in-vitro cellular uptake and localization. Thiophenol was used as molecular reporter and was bound to the Au surface by a chemisorption process in aqueous solution. The obtained colloidal solution was highly stable and no aggregation of the single nanospheres into larger clusters was observed. The nanoparticles were incubated in human prostatic cells with the aim of developing a robust, SERS-based method to differentiate normal and tumor cell lines. SERS imaging experiments showed that tumor cells uptake considerably larger amounts of nanoparticles in comparison to normal cells (up to 950% more); significant differences were also observed in the uptake kinetics. This largely different behaviour might be exploited in diagnostic and therapeutic applications.
Keywords: SERS; biosensing; nanomaterials; prostate cancer; single cell spectroscopy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Yuan Y., Panwar N., Yap S.H.K., Wu Q., Zeng S., Xu J., Tjin S.C., Song J., Qu J., Yong K.-T. SERS-based ultrasensitive sensing platform: An insight into design and practical applications. Coord. Chem. Rev. 2017;337:1–33. doi: 10.1016/j.ccr.2017.02.006. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

