Effectiveness of Subcutaneously Administered Leuprolide Acetate to Achieve Low Nadir Testosterone in Prostate Cancer Patients
- PMID: 30288142
- PMCID: PMC6168320
- DOI: 10.3909/riu0798
Effectiveness of Subcutaneously Administered Leuprolide Acetate to Achieve Low Nadir Testosterone in Prostate Cancer Patients
Erratum in
-
Correction for "Effectiveness of Subcutaneously Administered Leuprolide Acetate to Achieve Low Nadir Testosterone in Prostate Cancer Patients".Rev Urol. 2019;21(2-3):144. Rev Urol. 2019. PMID: 31768146 Free PMC article.
Abstract
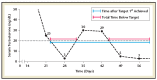
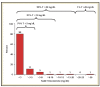
Evidence suggests lower nadir testosterone levels during the first year of androgen deprivation therapy improve advanced prostate cancer clinical outcomes. We evaluated pivotal trials for subcutaneously administered leuprolide acetate (1-, 3-, 4-, and 6-month doses) to determine nadir testosterone levels. Pooled analysis showed 99%, 97%, and 91% of patients reached nadir testosterone ≤20, ≤10, and ≤5 ng/dL respectively (median ≤3 ng/dL). Across all available categories, $88% of patients reached nadir testosterone ≤5 ng/dL, and <3% experienced a microsurge. Achievement and maintenance of low nadir testosterone levels may improve progression-free survival and time to onset of castrate-resistant prostate cancer.
Keywords: Androgen deprivation therapy; LHRH agonist; Leuprolide acetate; Nadir testosterone; Prostate cancer.
Figures

References
-
- Perachino M, Cavalli V, Bravi F. Testosterone levels in patients with metastatic prostate cancer treated with luteinizing hormone-releasing hormone therapy: prognostic significance? BJU Int. 2010;105:648–651. - PubMed
-
- Huggins C, Hodges CV. Studies on prostatic cancer. I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. AACR. 1941;1:293–297. - PubMed
-
- Oefelein MG, Feng A, Scolieri MJ, et al. Reassessment of the definition of castrate levels of testosterone: implications for clinical decision making. Urology. 2000;56:1021–1024. - PubMed
-
- van der Sluis TM, Bui HN, Meuleman EJ, et al. Lower testosterone levels with luteinizing hormone-releasing hormone agonist therapy than with surgical castration: new insights attained by mass spectrometry. J Urol. 2012;187:1601–1606. - PubMed
LinkOut - more resources
Full Text Sources
