Synthesis of novel perillyl-dihydropyrimidinone hybrids designed for antiproliferative activity
- PMID: 30288229
- PMCID: PMC6151448
- DOI: 10.1039/c8md00270c
Synthesis of novel perillyl-dihydropyrimidinone hybrids designed for antiproliferative activity
Abstract
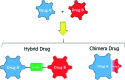
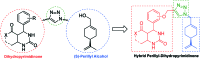
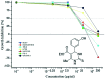
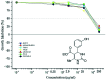
A series of fifteen novel dihydropyrimidinone hybrid compounds were synthesized in good yields via a multicomponent reaction combined with the Huisgen reaction. The antiproliferative activity was investigated against nine tumor cell lines, and four hybrid compounds (TGI < 10 μM) showed promising antiproliferative activity against the tumor cell lines OVCAR-3 (ovarian), UACC-62 (melanoma) and U251 (glioma). Several hybrid compounds assayed have high TGI values (TGI 147.92-507.82) for the human keratinocyte cell line (HaCat), which reveals selectivity to cancer cells.
Figures


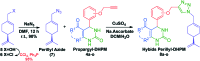


Similar articles
-
Synthesis and Investigation of the Role of Benzopyran Dihydropyrimidinone Hybrids in Cell Proliferation, Migration and Tumor Growth.Anticancer Agents Med Chem. 2019;19(2):276-288. doi: 10.2174/1871520618666180903101422. Anticancer Agents Med Chem. 2019. PMID: 30179143
-
Fe3O4 Nanoparticles Mediated Synthesis of Novel Isatin-dihydropyrimidinone Hybrid Molecules as Antioxidant and Cytotoxic Agents.Anticancer Agents Med Chem. 2017;17(3):456-463. doi: 10.2174/1871520616666160513132130. Anticancer Agents Med Chem. 2017. PMID: 27173964
-
Antiproliferative effect of synthetic cyclic imides (methylphtalimides, carboxylic acid phtalimides and itaconimides) against human cancer cell lines.Z Naturforsch C J Biosci. 2016 Nov 1;71(11-12):423-427. doi: 10.1515/znc-2016-0067. Z Naturforsch C J Biosci. 2016. PMID: 27768587
-
Bioactivity-guided isolation of cytotoxic sesquiterpene lactones of Gochnatia polymorpha ssp. floccosa.Phytother Res. 2012 Jul;26(7):1053-6. doi: 10.1002/ptr.3693. Epub 2011 Dec 22. Phytother Res. 2012. PMID: 22807254
-
Hybrids of 4-hydroxy derivatives of goniothalamin and piplartine bearing a diester or a 1,2,3-triazole linker as antiproliferative agents.Bioorg Chem. 2021 Nov;116:105292. doi: 10.1016/j.bioorg.2021.105292. Epub 2021 Aug 25. Bioorg Chem. 2021. PMID: 34509797
Cited by
-
Challenging the Biginelli scaffold to surpass the first line antitubercular drugs: Mycobacterium tuberculosis thymidine monophosphate kinase (TMPKmt) inhibition activity and molecular modelling studies.J Enzyme Inhib Med Chem. 2024 Dec;39(1):2386668. doi: 10.1080/14756366.2024.2386668. Epub 2024 Sep 11. J Enzyme Inhib Med Chem. 2024. PMID: 39258667 Free PMC article.
-
A comparative study between Cu(INA)2-MOF and [Cu(INA)2(H2O)4] complex for a click reaction and the Biginelli reaction under solvent-free conditions.RSC Adv. 2020 Jan 21;10(6):3407-3415. doi: 10.1039/c9ra10171c. eCollection 2020 Jan 16. RSC Adv. 2020. PMID: 35497731 Free PMC article.
-
A novel EGFR inhibitor, HNPMI, regulates apoptosis and oncogenesis by modulating BCL-2/BAX and p53 in colon cancer.Br J Pharmacol. 2024 Jan;181(1):107-124. doi: 10.1111/bph.16141. Epub 2023 Sep 12. Br J Pharmacol. 2024. PMID: 37183661 Free PMC article.
-
The Monoterpenoid Perillyl Alcohol: Anticancer Agent and Medium to Overcome Biological Barriers.Pharmaceutics. 2021 Dec 16;13(12):2167. doi: 10.3390/pharmaceutics13122167. Pharmaceutics. 2021. PMID: 34959448 Free PMC article. Review.
-
A Review on Recent Advances in Nitrogen-Containing Molecules and Their Biological Applications.Molecules. 2020 Apr 20;25(8):1909. doi: 10.3390/molecules25081909. Molecules. 2020. PMID: 32326131 Free PMC article. Review.
References
-
- Zhang H. Y. FEBS Lett. 2005;579:5260. - PubMed
-
- Maggiora G. M. J. J. Comput.-Aided Mol. Des. 2011;25:699. - PubMed
-
- Bansal Y., Silakari O. Eur. J. Med. Chem. 2014;76:31. - PubMed
-
- Youdim M. B. H., Buccafusco J. J. Trends Pharmacol. Sci. 2005;26:27. - PubMed
-
- Keith C. T., Borisy A. A., Stockwell B. R., Medina-Franco J. L., Giulianotti M. A., Welmaker G. S., Houghten R. A. Nat. Rev. Drug Discovery. Drug Discovery Today. 2005;2013;418:71. 495. - PubMed
LinkOut - more resources
Full Text Sources

