Differences between FLIM phasor analyses for data collected with the Becker and Hickl SPC830 card and with the FLIMbox card
- PMID: 30295346
- PMCID: PMC6240382
- DOI: 10.1002/jemt.23061
Differences between FLIM phasor analyses for data collected with the Becker and Hickl SPC830 card and with the FLIMbox card
Abstract
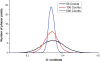
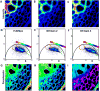
The phasor approach to FLIM (Fluorescence Lifetime Imaging Microscopy) is becoming popular due to the powerful fit free analysis and the visualization of the decay at each point in images of cells and tissues. However, although several implementation of the method are offered by manufactures of FLIM accessories for microscopes, the details of the conversion of the decay to phasors at each point in an image requires some consideration. Here, we show that if the decay is not properly acquired, the apparently simple phasor transformation can provide incorrect phasor plots and the results may be misinterpreted. In particular, we show the disagreement in experimental data acquired on the same samples using the two cards (FLIMbox, frequency domain and Becker & Hickl BH 830, time domain) and the effect produced by using the BH 830 card with different settings. This difference in data acquisition translates to the assignment of phasor components calculated using different acquisition parameters. This effect is already present in the original data that are not acquired with the proper parameters for the phasor conversion. We also show that the difference in the resolution of components already exists in the data acquired in the time domain when used with settings that do not allow acquisition of the fluorescence decay on a sufficient large time scale. RESEARCH HIGHLIGHTS: This paper is intended to made researchers aware of some simple requirements for the conversion of time-domain data (typically TCSPC) to phasors. The use of phasors for FLIM analysis has seen a surge of popularity. Since the phasor approach is a fit free method and has a powerful visualization of the data, it appears very simple to use. This paper shows that when the original data in the time domain is not acquired with the proper time range to cover the lifetimes in a sample, the conversion to phasors can produce very erroneous results. These results are appearing more frequently in the literature since many of the manufacturers of FLIM accessories for microscopes are now offering the phasor analysis in their software. Here, we show that the phasor transformation per se cannot correct for the problems with data acquisition and that one is misled to think that the "phasor approach" is a universal fix for the lack of the proper time range for data acquisition.
Keywords: Becker & Hickl; FLIM; FLIMBox; fluorescence; lifetime; phasor analysis.
© 2018 Wiley Periodicals, Inc.
Figures
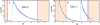

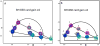

Similar articles
-
Fit-free analysis of fluorescence lifetime imaging data using the phasor approach.Nat Protoc. 2018 Sep;13(9):1979-2004. doi: 10.1038/s41596-018-0026-5. Nat Protoc. 2018. PMID: 30190551
-
FLUTE: A Python GUI for interactive phasor analysis of FLIM data.Biol Imaging. 2023 Nov 6;3:e21. doi: 10.1017/S2633903X23000211. eCollection 2023. Biol Imaging. 2023. PMID: 38487690 Free PMC article.
-
Label-free identification and differentiation of different microplastics using phasor analysis of fluorescence lifetime imaging microscopy (FLIM)-generated data.Chem Biol Interact. 2021 Jun 1;342:109466. doi: 10.1016/j.cbi.2021.109466. Epub 2021 Apr 15. Chem Biol Interact. 2021. PMID: 33865829
-
Linear Combination Properties of the Phasor Space in Fluorescence Imaging.Sensors (Basel). 2022 Jan 27;22(3):999. doi: 10.3390/s22030999. Sensors (Basel). 2022. PMID: 35161742 Free PMC article. Review.
-
The Phasor Plot: A Universal Circle to Advance Fluorescence Lifetime Analysis and Interpretation.Annu Rev Biophys. 2021 May 6;50:575-593. doi: 10.1146/annurev-biophys-062920-063631. Annu Rev Biophys. 2021. PMID: 33957055 Review.
Cited by
-
Altered substrate metabolism in neurodegenerative disease: new insights from metabolic imaging.J Neuroinflammation. 2021 Oct 28;18(1):248. doi: 10.1186/s12974-021-02305-w. J Neuroinflammation. 2021. PMID: 34711251 Free PMC article. Review.
-
Advances in fluorescence microscopy techniques to study kidney function.Nat Rev Nephrol. 2021 Feb;17(2):128-144. doi: 10.1038/s41581-020-00337-8. Epub 2020 Sep 18. Nat Rev Nephrol. 2021. PMID: 32948857 Review.
-
Quantitative melanoma diagnosis using spectral phasor analysis of hyperspectral imaging from label-free slices.Front Oncol. 2023 Nov 17;13:1296826. doi: 10.3389/fonc.2023.1296826. eCollection 2023. Front Oncol. 2023. PMID: 38162497 Free PMC article.
-
Deciphering amyloid fibril molecular maturation through FLIM-phasor analysis of thioflavin T.Biophys Rep (N Y). 2024 Feb 1;4(1):100145. doi: 10.1016/j.bpr.2024.100145. eCollection 2024 Mar 13. Biophys Rep (N Y). 2024. PMID: 38404533 Free PMC article.
-
Blind Resolution of Lifetime Components in Individual Pixels of Fluorescence Lifetime Images Using the Phasor Approach.J Phys Chem B. 2020 Nov 12;124(45):10126-10137. doi: 10.1021/acs.jpcb.0c06946. Epub 2020 Nov 3. J Phys Chem B. 2020. PMID: 33140960 Free PMC article.
References
-
- Becker W, Bergmann A, Haustein E, Petrasek Z, Schwille P, Biskup C, Kelbauskas L, Benndorf K, Klocker N, Anhut T, Riemann I, Konig K. Fluorescence lifetime images and correlation spectra obtained by multidimensional time-correlated single photon counting. Microsc Res Tech. 2006;69(3):186–95. - PubMed
-
- Becker W, Hirvonen LM, Milnes J, Conneely T, Jagutzki O, Netz H, Smietana S, Suhling K. A wide-field TCSPC FLIM system based on an MCP PMT with a delay-line anode. Rev Sci Instrum. 2016;87(9):093710. - PubMed

