Ligand binding properties of two Brugia malayi fatty acid and retinol (FAR) binding proteins and their vaccine efficacies against challenge infection in gerbils
- PMID: 30296268
- PMCID: PMC6193737
- DOI: 10.1371/journal.pntd.0006772
Ligand binding properties of two Brugia malayi fatty acid and retinol (FAR) binding proteins and their vaccine efficacies against challenge infection in gerbils
Abstract
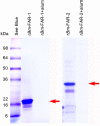
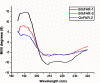
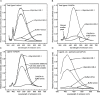
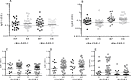
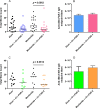
Parasitic nematodes produce an unusual class of fatty acid and retinol (FAR)-binding proteins that may scavenge host fatty acids and retinoids. Two FARs from Brugia malayi (Bm-FAR-1 and Bm-FAR-2) were expressed as recombinant proteins, and their ligand binding, structural characteristics, and immunogenicities examined. Circular dichroism showed that rBm-FAR-1 and rBm-FAR-2 are similarly rich in α-helix structure. Unexpectedly, however, their lipid binding activities were found to be readily differentiated. Both FARs bound retinol and cis-parinaric acid similarly, but, while rBm-FAR-1 induced a dramatic increase in fluorescence emission and blue shift in peak emission by the fluorophore-tagged fatty acid (dansyl-undecanoic acid), rBm-FAR-2 did not. Recombinant forms of the related proteins from Onchocerca volvulus, rOv-FAR-1 and rOv-FAR-2, were found to be similarly distinguishable. This is the first FAR-2 protein from parasitic nematodes that is being characterized. The relative protein abundance of Bm-FAR-1 was higher than Bm-FAR-2 in the lysates of different developmental stages of B. malayi. Both FAR proteins were targets of strong IgG1, IgG3 and IgE antibody in infected individuals and individuals who were classified as endemic normal or putatively immune. In a B. malayi infection model in gerbils, immunization with rBm-FAR-1 and rBm-FAR-2 formulated in a water-in-oil-emulsion (®Montanide-720) or alum elicited high titers of antigen-specific IgG, but only gerbils immunized with rBm-FAR-1 formulated with the former produced a statistically significant reduction in adult worms (68%) following challenge with B. malayi infective larvae. These results suggest that FAR proteins may play important roles in the survival of filarial nematodes in the host, and represent potential candidates for vaccine development against lymphatic filariasis and related filarial infections.
Conflict of interest statement
The authors declare that they have no competing interests, financial or otherwise.
Figures

Similar articles
-
Vaccination of Gerbils with Bm-103 and Bm-RAL-2 Concurrently or as a Fusion Protein Confers Consistent and Improved Protection against Brugia malayi Infection.PLoS Negl Trop Dis. 2016 Apr 5;10(4):e0004586. doi: 10.1371/journal.pntd.0004586. eCollection 2016 Apr. PLoS Negl Trop Dis. 2016. PMID: 27045170 Free PMC article.
-
Immunoprophylactic evaluation of a 37-kDa Brugia malayi recombinant antigen in lymphatic filariasis.Clin Microbiol Infect. 2006 Apr;12(4):361-8. doi: 10.1111/j.1469-0691.2006.01362.x. Clin Microbiol Infect. 2006. PMID: 16524413
-
Large extracellular loop of tetraspanin as a potential vaccine candidate for filariasis.PLoS One. 2013 Oct 11;8(10):e77394. doi: 10.1371/journal.pone.0077394. eCollection 2013. PLoS One. 2013. PMID: 24146990 Free PMC article.
-
Immune evasion genes from filarial nematodes.Int J Parasitol. 2001 Jul;31(9):889-98. doi: 10.1016/s0020-7519(01)00213-2. Int J Parasitol. 2001. PMID: 11406138 Review.
-
Future prospects and challenges of vaccines against filariasis.Parasite Immunol. 2012 May;34(5):243-53. doi: 10.1111/j.1365-3024.2011.01350.x. Parasite Immunol. 2012. PMID: 22150082 Review.
Cited by
-
De novo transcriptome reveals blood coagulation/antithrombin factors and infection mechanisms in Angiostrongylus cantonensis adult worms.Parasitology. 2021 Jun;148(7):857-870. doi: 10.1017/S0031182021000469. Epub 2021 Mar 17. Parasitology. 2021. PMID: 33729108 Free PMC article.
-
Promising Technologies in the Field of Helminth Vaccines.Front Immunol. 2021 Aug 19;12:711650. doi: 10.3389/fimmu.2021.711650. eCollection 2021. Front Immunol. 2021. PMID: 34489961 Free PMC article. Review.
-
Advancing a Human Onchocerciasis Vaccine From Antigen Discovery to Efficacy Studies Against Natural Infection of Cattle With Onchocerca ochengi.Front Cell Infect Microbiol. 2022 Apr 4;12:869039. doi: 10.3389/fcimb.2022.869039. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35444961 Free PMC article. Review.
-
Preliminary evaluations of 3-dimensional human skin models for their ability to facilitate in vitro the long-term development of the debilitating obligatory human parasite Onchocerca volvulus.PLoS Negl Trop Dis. 2020 Nov 5;14(11):e0008503. doi: 10.1371/journal.pntd.0008503. eCollection 2020 Nov. PLoS Negl Trop Dis. 2020. PMID: 33151944 Free PMC article.
-
Proteomic characterization and comparison of the infective and adult life stage secretomes from Necator americanus and Ancylostoma ceylanicum.PLoS Negl Trop Dis. 2025 Jan 20;19(1):e0012780. doi: 10.1371/journal.pntd.0012780. eCollection 2025 Jan. PLoS Negl Trop Dis. 2025. PMID: 39832284 Free PMC article.
References
-
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1211–59. Epub 2017/09/19. 10.1016/S0140-6736(17)32154-2 ; PubMed Central PMCID: PMC5605509. - DOI - PMC - PubMed
-
- Garofalo A, Klager SL, Rowlinson MC, Nirmalan N, Klion A, Allen JE, et al. The FAR proteins of filarial nematodes: secretion, glycosylation and lipid binding characteristics. Molecular and biochemical parasitology. 2002;122(2):161–70. Epub 2002/07/11. . - PubMed
-
- McDermott L, Cooper A, Kennedy MW. Novel classes of fatty acid and retinol binding protein from nematodes. Molecular and cellular biochemistry. 1999;192(1–2):69–75. Epub 1999/05/20. . - PubMed
-
- Nirmalan N, Cordeiro NJ, Klager SL, Bradley JE, Allen JE. Comparative analysis of glycosylated and nonglycosylated filarial homologues of the 20-kilodalton retinol binding protein from Onchocerca volvulus (Ov20). Infection and immunity. 1999;67(12):6329–34. Epub 1999/11/24. ; PubMed Central PMCID: PMC97037. - PMC - PubMed
-
- Kennedy MW, Garside LH, Goodrick LE, McDermott L, Brass A, Price NC, et al. The Ov20 protein of the parasitic nematode Onchocerca volvulus. A structurally novel class of small helix-rich retinol-binding proteins. The Journal of biological chemistry. 1997;272(47):29442–8. Epub 1997/12/31. . - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

