Input-specific maturation of NMDAR-mediated transmission onto parvalbumin-expressing interneurons in layers 2/3 of the visual cortex
- PMID: 30303753
- PMCID: PMC6337035
- DOI: 10.1152/jn.00495.2018
Input-specific maturation of NMDAR-mediated transmission onto parvalbumin-expressing interneurons in layers 2/3 of the visual cortex
Abstract
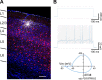
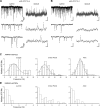
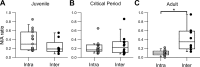
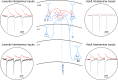
Parvalbumin-expressing (PV) GABAergic interneurons regulate local circuit dynamics. In terms of the excitation driving PV interneuron activity, the N-methyl-d-aspartate receptor (NMDAR)-mediated component onto PV interneurons tends to be smaller than that onto pyramidal neurons but makes a significant contribution to their physiology and development. In the visual cortex, PV interneurons mature during the critical period. We hypothesize that during the critical period, the NMDAR-mediated signaling and functional properties of glutamatergic synapses onto PV interneurons are developmentally regulated. We therefore compared the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR)- and NMDAR-mediated synaptic responses before (postnatal days 15-20, P15-P20), during (P25-P40), and after (P50-P60) the visual critical period. AMPAR miniature excitatory postsynaptic currents (mEPSCs) showed a developmental decrease in frequency, whereas NMDAR mEPSCs were absent or showed extremely low frequencies throughout development. For evoked responses, we consistently saw a NMDAR-mediated component, suggesting pre- or postsynaptic differences between evoked and spontaneous neurotransmission. Evoked responses showed input-specific developmental changes. For intralaminar inputs, the NMDAR-mediated component significantly decreased with development. This resulted in adult intralaminar inputs almost exclusively mediated by AMPARs, suited for the computation of synaptic inputs with precise timing, and likely having NMDAR-independent forms of plasticity. In contrast, interlaminar inputs maintained a stable NMDAR-mediated component throughout development but had a shift in the AMPAR paired-pulse ratio from depression to facilitation. Adult interlaminar inputs with facilitating AMPAR responses and a substantial NMDAR component would favor temporal integration of synaptic responses and could be modulated by NMDAR-dependent forms of plasticity. NEW & NOTEWORTHY We show for the first time input-specific developmental changes in the N-methyl-d-aspartate receptor component and short-term plasticity of the excitatory drive onto layers 2/3 parvalbumin-expressing (PV) interneurons in the visual cortex during the critical period. These developmental changes would lead to functionally distinct adult intralaminar and interlaminar glutamatergic inputs that would engage PV interneuron-mediated inhibition differently.
Keywords: AMPA receptors; NMDA receptors; PV interneurons.
Figures




References
-
- Abrahamsson T, Chou CY, Li SY, Mancino A, Costa RP, Brock JA, Nuro E, Buchanan KA, Elgar D, Blackman AV, Tudor-Jones A, Oyrer J, Farmer WT, Murai KK, Sjöström PJ. Differential regulation of evoked and spontaneous release by presynaptic NMDA receptors. Neuron 96: 839–855.e5, 2017. doi: 10.1016/j.neuron.2017.09.030. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

