Metal tolerance protein MTP6 affects mitochondrial iron and manganese homeostasis in cucumber
- PMID: 30304441
- PMCID: PMC6305187
- DOI: 10.1093/jxb/ery342
Metal tolerance protein MTP6 affects mitochondrial iron and manganese homeostasis in cucumber
Erratum in
-
Corrigendum to: Metal tolerance protein MTP6 affects mitochondrial iron and manganese homeostasis in cucumber.J Exp Bot. 2019 Jan 1;70(1):369. doi: 10.1093/jxb/ery399. J Exp Bot. 2019. PMID: 30481330 Free PMC article. No abstract available.
Retraction in
-
RETRACTION NOTICE.J Exp Bot. 2020 Oct 7;71(19):6187. doi: 10.1093/jxb/erz449. J Exp Bot. 2020. PMID: 31972017 Free PMC article. No abstract available.
Abstract
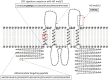
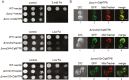
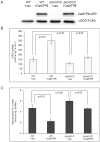
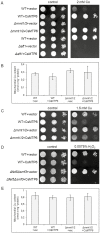
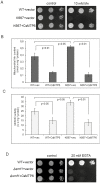
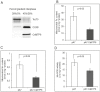
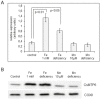
Members of the cation diffusion facilitator (CDF) family have been identified in all kingdoms of life. They have been divided into three subgroups, namely Zn-CDF, Fe/Zn-CDF, and Mn-CDF, based on their putative specificity to transported metal ions. The plant metal tolerance protein 6 (MTP6) proteins fall into the Fe/Zn-CDF subgroup; however, their function in iron/zinc transport has not yet been confirmed. Here, we characterized the MTP6 protein from cucumber, Cucumis sativus. When expressed in yeast and in protoplasts isolated from Arabidopsis cells, CsMTP6 localized in mitochondria and contributed to the efflux of Fe and Mn from these organelles. Immunolocalization of CsMTP6 in cucumber membranes confirmed this association with mitochondria. Root expression and protein levels of CsMTP6 were significantly up-regulated in conditions of Fe deficiency and excess, but were not affected by Mn availability. These results indicate that MTP6 proteins contribute to the distribution of Fe and Mn between the cytosol and mitochondria of plant cells, and are regulated by Fe to maintain mitochondrial and cytosolic iron homeostasis under varying conditions of Fe availability.
Figures

References
-
- Arrivault S, Senger T, Krämer U. 2006. The Arabidopsis metal tolerance protein AtMTP3 maintains metal homeostasis by mediating Zn exclusion from the shoot under Fe deficiency and Zn oversupply. The Plant Journal 46, 861–879. - PubMed
-
- Asada K, Takahashi M, Nagate M. 1974. Assay and inhibitors of spinach superoxide dismutase. Agricultural and Biological Chemistry 38, 471–473.
-
- Bradford MM. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry 72, 248–254. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

