Surface Modification of Magnetic Iron Oxide Nanoparticles
- PMID: 30304823
- PMCID: PMC6215286
- DOI: 10.3390/nano8100810
Surface Modification of Magnetic Iron Oxide Nanoparticles
Abstract
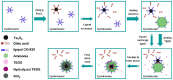
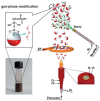
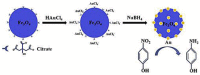
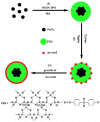
Functionalized iron oxide nanoparticles (IONPs) are of great interest due to wide range applications, especially in nanomedicine. However, they face challenges preventing their further applications such as rapid agglomeration, oxidation, etc. Appropriate surface modification of IONPs can conquer these barriers with improved physicochemical properties. This review summarizes recent advances in the surface modification of IONPs with small organic molecules, polymers and inorganic materials. The preparation methods, mechanisms and applications of surface-modified IONPs with different materials are discussed. Finally, the technical barriers of IONPs and their limitations in practical applications are pointed out, and the development trends and prospects are discussed.
Keywords: iron oxide nanoparticles; magnetic nanoparticles; nanomedicine; surface modification.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Recent progress on magnetic iron oxide nanoparticles: synthesis, surface functional strategies and biomedical applications.Sci Technol Adv Mater. 2015 Apr 28;16(2):023501. doi: 10.1088/1468-6996/16/2/023501. eCollection 2015 Apr. Sci Technol Adv Mater. 2015. PMID: 27877761 Free PMC article. Review.
-
Surface engineering of magnetic iron oxide nanoparticles by polymer grafting: synthesis progress and biomedical applications.Nanoscale. 2020 Jul 23;12(28):14957-14975. doi: 10.1039/d0nr03346d. Nanoscale. 2020. PMID: 32648868 Review.
-
Multifunctional Iron Oxide Magnetic Nanoparticles for Biomedical Applications: A Review.Materials (Basel). 2022 Jan 10;15(2):503. doi: 10.3390/ma15020503. Materials (Basel). 2022. PMID: 35057223 Free PMC article. Review.
-
Recent trends in preparation and biomedical applications of iron oxide nanoparticles.J Nanobiotechnology. 2024 Jan 8;22(1):24. doi: 10.1186/s12951-023-02235-0. J Nanobiotechnology. 2024. PMID: 38191388 Free PMC article. Review.
-
Unique Properties of Surface-Functionalized Nanoparticles for Bio-Application: Functionalization Mechanisms and Importance in Application.Nanomaterials (Basel). 2022 Apr 13;12(8):1333. doi: 10.3390/nano12081333. Nanomaterials (Basel). 2022. PMID: 35458041 Free PMC article. Review.
Cited by
-
Influence of Physicochemical Properties of Iron Oxide Nanoparticles on Their Antibacterial Activity.ACS Omega. 2024 Jul 25;9(31):33303-33334. doi: 10.1021/acsomega.4c02822. eCollection 2024 Aug 6. ACS Omega. 2024. PMID: 39130596 Free PMC article. Review.
-
Magnetic iron oxides nanocomposites: synthetic techniques and environmental applications for wastewater treatment.Discov Nano. 2024 Sep 28;19(1):158. doi: 10.1186/s11671-024-04102-9. Discov Nano. 2024. PMID: 39342049 Free PMC article. Review.
-
Polyethylene Glycol-Chitosan Oligosaccharide-Coated Superparamagnetic Iron Oxide Nanoparticles: A Novel Drug Delivery System for Curcumin Diglutaric Acid.Biomolecules. 2020 Jan 2;10(1):73. doi: 10.3390/biom10010073. Biomolecules. 2020. PMID: 31906490 Free PMC article.
-
Urinary PCA3 detection in prostate cancer by magnetic nanoparticles coupled with colorimetric enzyme-linked oligonucleotide assay.EXCLI J. 2020 Apr 15;19:501-513. doi: 10.17179/excli2020-1036. eCollection 2020. EXCLI J. 2020. PMID: 32398974 Free PMC article.
-
Design and preparation of amino-functionalized core-shell magnetic nanoparticles for photocatalytic application and investigation of cytotoxicity effects.J Environ Health Sci Eng. 2022 Nov 30;21(1):93-105. doi: 10.1007/s40201-022-00842-x. eCollection 2023 Jun. J Environ Health Sci Eng. 2022. PMID: 37159737 Free PMC article.
References
-
- Rossi L.M., Costa N.J.S., Silva F.P., Wojcieszak R. Magnetic nanomaterials in catalysis: Advanced catalysts for magnetic separation and beyond. Green Chem. 2014;16:2906–2933. doi: 10.1039/c4gc00164h. - DOI
-
- Zhang H.W., Liu Y., Sun S.H. Synthesis and assembly of magnetic nanoparticles for information and energy storage applications. Front. Phys. Chin. 2010;5:347–356. doi: 10.1007/s11467-010-0104-9. - DOI
-
- Li X.-B., Gao Y.-J., Wang Y., Zhan F., Zhang X.-Y., Kong Q.-Y., Zhao N.-J., Guo Q., Wu H.-L., Li Z.-J., et al. Self-Assembled Framework Enhances Electronic Communication of Ultrasmall-Sized Nanoparticles for Exceptional Solar Hydrogen Evolution. J. Am. Chem. Soc. 2017;139:4789–4796. doi: 10.1021/jacs.6b12976. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

