Nutritional Modulation of AMPK-Impact upon Metabolic-Inflammation
- PMID: 30304866
- PMCID: PMC6213547
- DOI: 10.3390/ijms19103092
Nutritional Modulation of AMPK-Impact upon Metabolic-Inflammation
Abstract
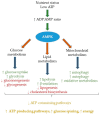
Nutritional status provides metabolic substrates to activate AMP-Activated Protein Kinase (AMPK), the energy sensor that regulates metabolism. Recent evidence has demonstrated that AMPK has wider functions with respect to regulating immune cell metabolism and function. One such example is the regulatory role that AMPK has on NLRP3-inlflammasome and IL-1β biology. This in turn can result in subsequent negative downstream effects on glucose, lipid and insulin metabolism. Nutrient stress in the form of obesity can impact AMPK and whole-body metabolism, leading to complications such as type 2 diabetes and cancer risk. There is a lack of data regarding the nature and extent that nutrient status has on AMPK and metabolic-inflammation. However, emerging work elucidates to a direct role of individual nutrients on AMPK and metabolic-inflammation, as a possible means of modulating AMPK activity. The posit being to use such nutritional agents to re-configure metabolic-inflammation towards more oxidative phosphorylation and promote the resolution of inflammation. The complex paradigm will be discussed within the context of if/how dietary components, nutrients including fatty acids and non-nutrient food components, such as resveratrol, berberine, curcumin and the flavonoid genistein, modulate AMPK dependent processes relating to inflammation and metabolism.
Keywords: AMPK; IL-1β; NLRP3; dietary fatty acids; metabolic-inflammation; nutrigenomics; nutrition.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Finucane O.M., Lyons C.L., Murphy A.M., Reynolds C.M., Klinger R., Healy N.P., Cooke A.A., Coll R.C., McAllan L., Nilaweera K.N., et al. Monounsaturated fatty acid-enriched high-fat diets impede adipose NLRP3 inflammasome-mediated IL-1? Secretion and insulin resistance despite obesity. Diabetes. 2015;64:2116–2128. doi: 10.2337/db14-1098. - DOI - PubMed
-
- Hawley S.A., Davison M., Woods A., Davies S.P., Beri R.K., Carling D., Hardie D.G. Characterization of the AMP-activated protein kinase kinase from rat liver and identification of threonine 172 as the major site at which it phosphorylates AMP-activated protein kinase. J. Biol. Chem. 1996;271:27879–27887. doi: 10.1074/jbc.271.44.27879. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

