Pullulan-Based Nanoparticle-HSA Complex Formation and Drug Release Influenced by Surface Charge
- PMID: 30306404
- PMCID: PMC6179976
- DOI: 10.1186/s11671-018-2729-5
Pullulan-Based Nanoparticle-HSA Complex Formation and Drug Release Influenced by Surface Charge
Abstract
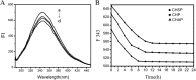
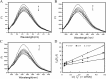
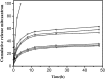
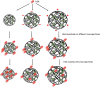
The nanomaterial composition of nanoparticles and their protein adsorption in the blood is of great significance in the design of drug-loaded nanoparticles. To explore the interaction between the different surface components of nanoparticles (NPs) and protein, we synthesized three kinds of pullulan NP polymers: cholesteric hydrophobically (CH) modified pullulan (CHP), CH-modified animated pullulan (CHAP), and CH-modified carboxylated pullulan (CHSP). Pullulan NPs were prepared by the dialysis method. Dynamic light scattering was used to determine the charge and size of the three NPs. The size of NPs was altered by the number of charge groups when polymers contain the same degree of cholesterol substitution. The zeta potentials were + 12.9, - 15.4, and - 0.698 mV for CHAP, CHSP, and CHP, respectively, and the dimensions were 116.9, 156.9, and 73.1 nm, respectively. Isothermal titration calorimetry was used to determine the thermodynamic changes of NPs with different surface charge, and the effect of human serum albumin (HSA) on the titration was investigated. The changes of enthalpy and entropy demonstrated an interaction between NPs and HSA; the binding constant (Kb) for CHSP, CHP, and CHAP was 1.41, 27.7, and 412 × 104 M-1, respectively, with the positive charge for CHAP-HSA, uncharged for CHP-HSA, and negative charge for CHSP-HSA complex. Fluorescence and circular dichroism spectroscopy were used to determine the protein structure change after the complexation between NPs and HSA. The NP and HSA complexation is a complicated process composed of protein α-helical content reduction and the peptide chain extension; CHP NPs had the largest reduction in HSA α-helical content. The drug release rates of all compounds of NP and HSA were significantly lower than those of free drug and drug-loaded NPs after 48 h. The highest and lowest rates were observed in CHSP-HSA and CHP-HSA, respectively. The drug release was significantly influenced by the adsorption of HSA on NPs, and the size and surface charge of NPs played an important role in this process.
Keywords: Binding constant; Drug release; Human serum albumin; Nanoparticles; α-Helix.
Conflict of interest statement
Competing Interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

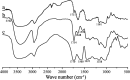


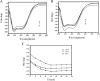
 ), CHAP NPs (
), CHAP NPs ( ), and CHSP NPs (
), and CHSP NPs ( )
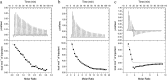
)
Similar articles
-
Effect of pullulan nanoparticle surface charges on HSA complexation and drug release behavior of HSA-bound nanoparticles.PLoS One. 2012;7(11):e49304. doi: 10.1371/journal.pone.0049304. Epub 2012 Nov 14. PLoS One. 2012. PMID: 23166632 Free PMC article.
-
Cholesterol-Modified Amino-Pullulan Nanoparticles as a Drug Carrier: Comparative Study of Cholesterol-Modified Carboxyethyl Pullulan and Pullulan Nanoparticles.Nanomaterials (Basel). 2016 Sep 8;6(9):165. doi: 10.3390/nano6090165. Nanomaterials (Basel). 2016. PMID: 28335293 Free PMC article.
-
Effects of Particle Hydrophobicity, Surface Charge, Media pH Value and Complexation with Human Serum Albumin on Drug Release Behavior of Mitoxantrone-Loaded Pullulan Nanoparticles.Nanomaterials (Basel). 2015 Dec 25;6(1):2. doi: 10.3390/nano6010002. Nanomaterials (Basel). 2015. PMID: 28344259 Free PMC article.
-
Synthesis and characterization of biotin modified cholesteryl pullulan as a novel anticancer drug carrier.Carbohydr Polym. 2014 Jan;99:720-7. doi: 10.1016/j.carbpol.2013.09.013. Epub 2013 Sep 10. Carbohydr Polym. 2014. PMID: 24274563
-
Surface engineering of inorganic nanoparticles for imaging and therapy.Adv Drug Deliv Rev. 2013 May;65(5):622-48. doi: 10.1016/j.addr.2012.08.015. Epub 2012 Sep 6. Adv Drug Deliv Rev. 2013. PMID: 22975010 Review.
Cited by
-
A Novel Delivery System of RGD-HSA Loaded GEM/CUR Nanoparticles for the Treatment of Pancreatic Cancer Therapy.Drug Des Devel Ther. 2022 Jul 26;16:2395-2406. doi: 10.2147/DDDT.S366558. eCollection 2022. Drug Des Devel Ther. 2022. PMID: 35923931 Free PMC article.
-
Sorafenib/2800Z Co-Loaded into Cholesterol and PEG Grafted Polylysine NPs for Liver Cancer Treatment.Pharmaceuticals (Basel). 2023 Jan 13;16(1):119. doi: 10.3390/ph16010119. Pharmaceuticals (Basel). 2023. PMID: 36678616 Free PMC article.
References
-
- Mody VV, et al. Magnetic nanoparticle drug delivery systems for targeting tumor. Appl Nanosci. 2014;4:385–392. doi: 10.1007/s13204-013-0216-y. - DOI
Grants and funding
- No. 81303254, NO: 81641140/National Natural Science Foundation of China
- No. WJ2017M214/Hubei Province health and family planning scientific research project
- No. 2016JJ2088/Natural Science Foundation of Hunan Province
- No.B2017073/the Health and Family planning commission of Hunan Province research project
LinkOut - more resources
Full Text Sources
Miscellaneous

