Protein kinase C and protein kinase A are involved in the protection of recombinant human glucagon-like peptide-1 on glomeruli and tubules in diabetic rats
- PMID: 30307132
- PMCID: PMC6497589
- DOI: 10.1111/jdi.12956
Protein kinase C and protein kinase A are involved in the protection of recombinant human glucagon-like peptide-1 on glomeruli and tubules in diabetic rats
Abstract
Aims/introduction: Blockade or reversal the progression of diabetic nephropathy is a clinical challenge. The aim of the present study was to examine whether recombinant human glucagon-like peptide-1 (rhGLP-1) has an effect on alleviating urinary protein and urinary albumin levels in diabetic rats.
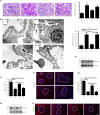
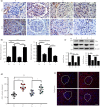
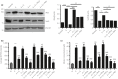
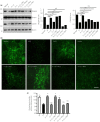
Materials and methods: Streptozotocin-induced diabetes rats were treated with rhGLP-1 insulin and saline. Using immunostaining, hematoxylin-eosin, electron microscopy and periodic acid-Schiff staining to study the pathology of diabetic nephropathy, and we carried out quantitative reverse transcription polymerase chain reaction, western blot and immunohistochemistry to identify the differentially expressed proteins. The mechanism was studied through advanced glycation end-products-induced tubular epithelial cells.
Results: rhGLP-1 inhibits protein kinase C (PKC)-β, but increases protein kinase A (PKA), which reduces oxidative stress in glomeruli and in cultured glomerular microvascular endothelial cells. In tubules, rhGLP-1 increased the expression of two key proteins related to re-absorption - megalin and cubilin - which was accompanied by downregulation of PKC-β and upregulation of PKA. On human proximal tubular epithelial cells, rhGLP-1 enhanced the absorption of albumin, and this was blocked by a PKC activator or PKA inhibitor.
Conclusions: These findings suggest that rhGLP-1 can reverse diabetic nephropathy by protecting both glomeruli and tubules by inhibiting PKC and activating PKA.
Keywords: Diabetic nephropathy; Protein kinase; Recombinant human glucagon-like peptide-1.
© 2018 The Authors. Journal of Diabetes Investigation published by Asian Association for the Study of Diabetes (AASD) and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Recombinant human GLP-1(rhGLP-1) alleviating renal tubulointestitial injury in diabetic STZ-induced rats.Biochem Biophys Res Commun. 2018 Jan 1;495(1):793-800. doi: 10.1016/j.bbrc.2017.11.076. Epub 2017 Nov 12. Biochem Biophys Res Commun. 2018. PMID: 29137984
-
Gliquidone decreases urinary protein by promoting tubular reabsorption in diabetic Goto-Kakizaki rats.J Endocrinol. 2014 Jan 8;220(2):129-41. doi: 10.1530/JOE-13-0199. Print 2014 Feb. J Endocrinol. 2014. PMID: 24254365
-
Glucagon-like peptide-1 receptor agonist inhibits asymmetric dimethylarginine generation in the kidney of streptozotocin-induced diabetic rats by blocking advanced glycation end product-induced protein arginine methyltranferase-1 expression.Am J Pathol. 2013 Jan;182(1):132-41. doi: 10.1016/j.ajpath.2012.09.016. Epub 2012 Nov 14. Am J Pathol. 2013. PMID: 23159951
-
Pathogenesis of diabetic nephropathy: the role of oxidative stress and protein kinase C.Diabetes Res Clin Pract. 1999 Sep;45(2-3):147-51. doi: 10.1016/s0168-8227(99)00044-3. Diabetes Res Clin Pract. 1999. PMID: 10588367 Review.
-
Advanced glycation endproducts and the kidney.Przegl Lek. 1998;55 Suppl 1:29-31. Przegl Lek. 1998. PMID: 9857688 Review. No abstract available.
Cited by
-
GLP-1 receptor agonists in diabetic kidney disease: current evidence and future directions.Kidney Res Clin Pract. 2022 Mar;41(2):136-149. doi: 10.23876/j.krcp.22.001. Epub 2022 Mar 25. Kidney Res Clin Pract. 2022. PMID: 35391537 Free PMC article.
-
The role of oxidative stress in diabetes mellitus-induced vascular endothelial dysfunction.Cardiovasc Diabetol. 2023 Sep 2;22(1):237. doi: 10.1186/s12933-023-01965-7. Cardiovasc Diabetol. 2023. PMID: 37660030 Free PMC article. Review.
-
Renoprotective Effects of DPP-4 Inhibitors.Antioxidants (Basel). 2021 Feb 5;10(2):246. doi: 10.3390/antiox10020246. Antioxidants (Basel). 2021. PMID: 33562528 Free PMC article. Review.
-
Recent evidence in the etiology and treatment for diabetic kidney disease.J Diabetes Investig. 2021 May;12(5):694-696. doi: 10.1111/jdi.13433. Epub 2020 Nov 15. J Diabetes Investig. 2021. PMID: 33031619 Free PMC article.
-
New prospects for incretin-related drugs in the treatment of type 2 diabetes.J Diabetes Investig. 2021 Jul;12(7):1141-1143. doi: 10.1111/jdi.13460. Epub 2020 Dec 9. J Diabetes Investig. 2021. PMID: 33151639 Free PMC article.
References
-
- Hsieh MC, Tien KJ, Perng DS, et al Diabetic nephropathy and risk factors for peripheral artery disease in Chinese with type 2 diabetes mellitus. Metabolism 2009; 58: 504–509. - PubMed
-
- Stanton RC, King GL. A complex interplay of factors causes diabetic nephropathy. Metabolism 2011; 60: 591–593. - PubMed
-
- Bhattacharjee N, Barma S, Konwar N, et al Mechanistic insight of diabetic nephropathy and its pharmacotherapeutic targets: an update. Eur J Pharmacol 2016; 791: 8–24. - PubMed
-
- Pofi R, Di Mario F, Gigante A, et al Diabetic nephropathy: focus on current and future therapeutic strategies. Curr Drug Metab 2016; 17: 497–502. - PubMed
-
- Lee YS, Jun HS. Anti‐diabetic actions of glucagon‐like peptide‐1 on pancreatic beta‐cells. Metabolism 2014; 63: 9–19. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

