Subtype Diversification and Synaptic Specificity of Stem Cell-Derived Spinal Interneurons
- PMID: 30308166
- PMCID: PMC6590086
- DOI: 10.1016/j.neuron.2018.09.016
Subtype Diversification and Synaptic Specificity of Stem Cell-Derived Spinal Interneurons
Abstract
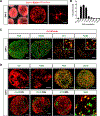
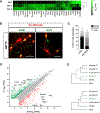
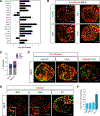
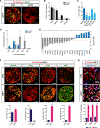
Neuronal diversification is a fundamental step in the construction of functional neural circuits, but how neurons generated from single progenitor domains acquire diverse subtype identities remains poorly understood. Here we developed an embryonic stem cell (ESC)-based system to model subtype diversification of V1 interneurons, a class of spinal neurons comprising four clades collectively containing dozens of molecularly distinct neuronal subtypes. We demonstrate that V1 subtype diversity can be modified by extrinsic signals. Inhibition of Notch and activation of retinoid signaling results in a switch to MafA clade identity and enriches differentiation of Renshaw cells, a specialized MafA subtype that mediates recurrent inhibition of spinal motor neurons. We show that Renshaw cells are intrinsically programmed to migrate to species-specific laminae upon transplantation and to form subtype-specific synapses with motor neurons. Our results demonstrate that stem cell-derived neuronal subtypes can be used to investigate mechanisms underlying neuronal subtype specification and circuit assembly.
Keywords: Renshaw cell; V1 interneuron; differentiation; embryonic stem cells; in vitro; motor neuron; neurons; spinal cord; subtype; synaptic specificity.
Copyright © 2018. Published by Elsevier Inc.
Figures


Comment in
-
Spinal Interneurons "à La Carte".Neuron. 2018 Oct 10;100(1):3-6. doi: 10.1016/j.neuron.2018.09.034. Neuron. 2018. PMID: 30308170
References
-
- Abbondanzo SJ, Gadi I, and Stewart CL (1993). Derivation of embryonic stem cell lines. Methods Enzymol. 225, 803–823. - PubMed
-
- Albuquerque C, Joseph DJ, Choudhury P, and MacDermott AB (2009). Dissection, plating, and maintenance of cortical astrocyte cultures. Cold Spring Harb. Protoc. 2009, pdb.prot5273. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

