The expanding world of biosynthetic pericyclases: cooperation of experiment and theory for discovery
- PMID: 30311924
- PMCID: PMC6461539
- DOI: 10.1039/c8np00075a
The expanding world of biosynthetic pericyclases: cooperation of experiment and theory for discovery
Abstract
Covering: 2000 to 2018 Pericyclic reactions are a distinct class of reactions that have wide synthetic utility. Before the recent discoveries described in this review, enzyme-catalyzed pericyclic reactions were not widely known to be involved in biosynthesis. This situation is changing rapidly. We define the scope of pericyclic reactions, give a historical account of their discoveries as biosynthetic reactions, and provide evidence that there are many enzymes in nature that catalyze pericyclic reactions. These enzymes, the "pericyclases," are the subject of this review.
Conflict of interest statement
Conflicts of interest
There are no conflicts to declare.
Figures


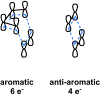









References
-
- Hoffmann R and Woodward RB, Acc. Chem. Res, 1968, 1, 17–22.
- Woodward RBand Hoffmann R, Angew. Chem. Int. Ed. Engl 1969, 8, 781; Verlag Chemie BmgH, Academic Press, 1970.
-
-
Ref. 1, page 169.
-
-
- Houk KN, Lin Y-T, and Brown FK, J. Am. Chem. Soc, 1986, 108, 554. - PubMed
-
- Beno BR, Houk KN, and Singleton DA, J. Am. Chem. Soc, 1996, 118, 9984–9985.
-
- Getty SJ and Borden WT, J. Am. Chem. Soc, 113, 4334–4335 (1991).

