GABAergic Interneurons in Seizures: Investigating Causality With Optogenetics
- PMID: 30317911
- PMCID: PMC6745605
- DOI: 10.1177/1073858418805002
GABAergic Interneurons in Seizures: Investigating Causality With Optogenetics
Abstract
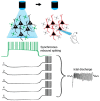
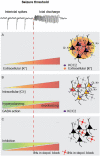
Seizures are complex pathological network events characterized by excessive and hypersynchronized activity of neurons, including a highly diverse population of GABAergic interneurons. Although the primary function of inhibitory interneurons under normal conditions is to restrain excitation in the brain, this system appears to fail intermittently, allowing runaway excitation. Recent developments in optogenetics, combined with genetic tools and advanced electrophysiological and imaging techniques, allow us for the first time to assess the causal roles of identified cell-types in network dynamics. While these methods have greatly increased our understanding of cortical microcircuits in epilepsy, the roles played by individual GABAergic cell-types in controlling ictogenesis remain incompletely resolved. Indeed, the ability of interneurons to suppress epileptic discharges varies across different subtypes, and an accumulating body of evidence paradoxically implicates some interneuron subtypes in the initiation and maintenance of epileptiform activity. Here, we bring together findings from this growing field and discuss what can be inferred regarding the causal role of different GABAergic cell-types in seizures.
Keywords: epilepsy; interictal spikes; interneurons; optogenetics; seizures.
Conflict of interest statement
Figures


References
-
- Avoli M, D’Antuono M, Louvel J, Köhling R, Biagini G, Pumain R, and others. 2002. Network and pharmacological mechanisms leading to epileptiform synchronization in the limbic system in vitro. Prog Neurobiol 68:167–207. - PubMed

