The Nontoxic Cholera B Subunit Is a Potent Adjuvant for Intradermal DC-Targeted Vaccination
- PMID: 30319653
- PMCID: PMC6171476
- DOI: 10.3389/fimmu.2018.02212
The Nontoxic Cholera B Subunit Is a Potent Adjuvant for Intradermal DC-Targeted Vaccination
Abstract
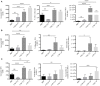
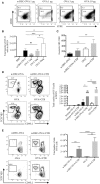
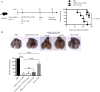
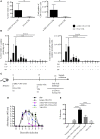
CD4+ T cells are major players in the immune response against several diseases; including AIDS, leishmaniasis, tuberculosis, influenza and cancer. Their activation has been successfully achieved by administering antigen coupled with antibodies, against DC-specific receptors in combination with adjuvants. Unfortunately, most of the adjuvants used so far in experimental models are unsuitable for human use. Therefore, human DC-targeted vaccination awaits the description of potent, yet nontoxic adjuvants. The nontoxic cholera B subunit (CTB) can be safely used in humans and it has the potential to activate CD4+ T cell responses. However, it remains unclear whether CTB can promote DC activation and can act as an adjuvant for DC-targeted antigens. Here, we evaluated the CTB's capacity to activate DCs and CD4+ T cell responses, and to generate long-lasting protective immunity. Intradermal (i.d.) administration of CTB promoted late and prolonged activation and accumulation of skin and lymphoid-resident DCs. When CTB was co-administered with anti-DEC205-OVA, it promoted CD4+ T cell expansion, differentiation, and infiltration to peripheral nonlymphoid tissues, i.e., the skin, lungs and intestine. Indeed, CTB promoted a polyfunctional CD4+ T cell response, including the priming of Th1 and Th17 cells, as well as resident memory T (RM) cell differentiation in peripheral nonlymphoid tissues. It is worth noting that CTB together with a DC-targeted antigen promoted local and systemic protection against experimental melanoma and murine rotavirus. We conclude that CTB administered i.d. can be used as an adjuvant to DC-targeted antigens for the induction of broad CD4+ T cell responses as well as for promoting long-lasting protective immunity.
Keywords: CTB; T cells; adjuvant; anti-DEC205; dendritic cells; memory; skin.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

