Techniques to stimulate and interrogate cell-cell adhesion mechanics
- PMID: 30320194
- PMCID: PMC6181239
- DOI: 10.1016/j.eml.2017.12.002
Techniques to stimulate and interrogate cell-cell adhesion mechanics
Abstract
Cell-cell adhesions maintain the mechanical integrity of multicellular tissues and have recently been found to act as mechanotransducers, translating mechanical cues into biochemical signals. Mechanotransduction studies have primarily focused on focal adhesions, sites of cell-substrate attachment. These studies leverage technical advances in devices and systems interfacing with living cells through cell-extracellular matrix adhesions. As reports of aberrant signal transduction originating from mutations in cell-cell adhesion molecules are being increasingly associated with disease states, growing attention is being paid to this intercellular signaling hub. Along with this renewed focus, new requirements arise for the interrogation and stimulation of cell-cell adhesive junctions. This review covers established experimental techniques for stimulation and interrogation of cell-cell adhesion from cell pairs to monolayers.
Keywords: BioMEMS; Cell mechanics; Cell–cell adhesion; FRET; Mechanobiology.
Figures
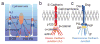
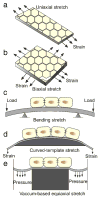
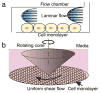
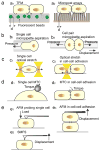


References
-
- Gumbiner BM. Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell. 1996;84:345–357. - PubMed
-
- Ingber DE. Cellular mechanotransduction: putting all the pieces together again. FASEB J. 2006;20:811–827. - PubMed
-
- Winograd-Katz SE, Fassler R, Geiger B, Legate KR. The integrin adhesome: from genes and proteins to human disease. Nat Rev Mol Cell Biol. 2014;15:273–288. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
