Impact of Next-generation Sequencing Defined Human Immunodeficiency Virus Pretreatment Drug Resistance on Virological Outcomes in the ANRS 12249 Treatment-as-Prevention Trial
- PMID: 30321314
- PMCID: PMC6603266
- DOI: 10.1093/cid/ciy881
Impact of Next-generation Sequencing Defined Human Immunodeficiency Virus Pretreatment Drug Resistance on Virological Outcomes in the ANRS 12249 Treatment-as-Prevention Trial
Abstract
Background: Previous studies in human immunodeficiency virus (HIV)-positive individuals on thymidine analogue backbone antiretroviral therapy (ART) with either nevirapine or efavirenz have suggested poorer virological outcomes in the presence of pretreatment drug resistance (PDR). We assessed the impact of PDR on virological suppression (VS; <50 copies/mL) in individuals prescribed primarily tenofovir/emtricitabine/efavirenz in rural KwaZulu-Natal within a treatment-as-prevention trial.
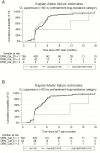
Methods: Among 1557 HIV-positive individuals who reported no prior ART at study entry and provided plasma samples, 1328 individuals with entry viral load (VL) >1000 copies/mL had next-generation sequencing (NGS) of the HIV pol gene with MiSeq technology. Results were obtained for 1148 individuals, and the presence of PDR was assessed at 5% and 20% detection thresholds. Virological outcome was assessed using Cox regression in 837 of 920 ART initiators with at least 1 follow-up VL after ART initiation.
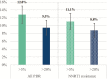
Results: PDR prevalence was 9.5% (109/1148) and 12.8% (147/1148) at 20% and 5% thresholds, respectively. After a median of 1.36 years (interquartile range, 0.91-2.13), mostly on fixed-dose combination tenofovir/emtricitabine/efavirenz, presence of both nonnucleoside reverse transcriptase inhibitor (NNRTI)/nucleoside reverse transcriptase inhibitor PDR vs no PDR was associated with longer time to VS (adjusted hazard ratio [aHR], 0.32; 95% confidence interval [CI], 0.12-0.86), while there was no difference between those with only NNRTI PDR vs no PDR (aHR, 1.05; 95% CI, 0.82-1.34) at the 5% threshold. Similar differences were observed for mutations detected at the 20% threshold, although without statistical significance.
Conclusions: NGS uncovered a high prevalence of PDR among participants enrolled in trial clinics in rural KwaZulu-Natal. Dual-class PDR to a mainly tenofovir/emtricitabine/efavirenz regimen was associated with poorer VS. However, there was no impact of NNRTI PDR alone.
Clinical trials tegistration: NCT01509508; South African National Clinical Trials Register: DOH-27-0512-3974.
Keywords: HIV; antiretroviral therapy; next-generation sequencing; pretreatment drug resistance; virological response.
© The Author(s) 2018. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures

Comment in
-
The Clinical Implications of Pretreatment Drug Resistance-A Moving Target.Clin Infect Dis. 2019 Jul 2;69(2):215-217. doi: 10.1093/cid/ciy895. Clin Infect Dis. 2019. PMID: 30321316 No abstract available.
-
Does Tenofovir-containing First-line Antiretroviral Therapy Mitigate the Impact of Pretreatment Non-nucleoside Reverse Transcriptase Inhibitor Drug Resistance?Clin Infect Dis. 2019 May 30;68(12):2158-2160. doi: 10.1093/cid/ciy1070. Clin Infect Dis. 2019. PMID: 30561519 No abstract available.
-
Is increasing pretreatment HIV drug resistance a real menace or minor detail? - Authors' reply.Lancet HIV. 2020 May;7(5):e317-e318. doi: 10.1016/S2352-3018(20)30049-7. Lancet HIV. 2020. PMID: 32386719 No abstract available.
References
-
- World Health Organization. Global AIDS Update 2016. Available at: http://www.who.int/hiv/pub/arv/global-AIDS-update-2016_en.pdf?ua=1. Accessed 06 October 2017.
-
- Boender TS, Hoenderboom BM, Sigaloff KC, et al. Pretreatment HIV drug resistance increases regimen switches in sub-Saharan Africa. Clin Infect Dis 2015; 61:1749–58. - PubMed
-
- Hamers RL, Schuurman R, Sigaloff KC, et al. ; PharmAccess African Studies to Evaluate Resistance Investigators Effect of pretreatment HIV-1 drug resistance on immunological, virological, and drug-resistance outcomes of first-line antiretroviral treatment in sub-Saharan Africa: a multicentre cohort study. Lancet Infect Dis 2012; 12:307–17. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

