Immune Stimulation of RAP domain binding protein (rTgRA15) from Toxoplasma gondii
- PMID: 30332344
- PMCID: PMC6300731
- DOI: 10.1080/20477724.2018.1536854
Immune Stimulation of RAP domain binding protein (rTgRA15) from Toxoplasma gondii
Abstract
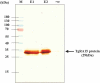
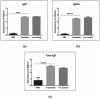
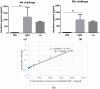
Toxoplasmosis, a parasitic disease in human and animals, is caused by Toxoplasma gondii. Our previous study has led to the discovery of a novel RAP domain binding protein antigen (TgRA15), an apparent in-vivo induced antigen recognised by antibodies in acutely infected individuals. This study is aimed to evaluate the humoral response and cytokine release elicited by recombinant TgRA15 protein in C57BL/6 mice, demonstrating its potential as a candidate vaccine for Toxoplasma gondii infection. In this study, the recombinant TgRA15 protein was expressed in Escherichia coli, purified and refolded into soluble form. C57BL/6 mice were immunised intradermally with the antigen and CASAC (Combined Adjuvant for Synergistic Activation of Cellular immunity). Antigen-specific humoral and cell-mediated responses were evaluated using Western blot and ELISA. The total IgG, IgG1 and IgG2a antibodies specific to the antigen were significantly increased in treatment group compare to control group. A higher level of interferon gamma (IFN-γ) secretion was demonstrated in the mice group receiving booster doses of rTgRA15 protein, suggesting a potential Th1-mediated response. In conclusion, the rTgRA15 protein has the potential to generate specific antibody response and elicit cellular response, thus potentially serve as a vaccine candidate against T. gondii infection.
Keywords: TgRA; Toxoplasmosis; antibody; immunity; interferon gamma; mice; vaccine.
Figures
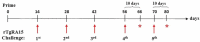
References
-
- Jongert E, Roberts CW, Gargano N, et al. Vaccines against Toxoplasma gondii: challenges and opportunities. Mem Inst Oswaldo Cruz. 2009March;104(2):252–266. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
