A 250 μm × 57 μm Microscale Opto-electronically Transduced Electrodes (MOTEs) for Neural Recording
- PMID: 30334768
- PMCID: PMC6338085
- DOI: 10.1109/TBCAS.2018.2876069
A 250 μm × 57 μm Microscale Opto-electronically Transduced Electrodes (MOTEs) for Neural Recording
Abstract
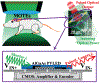
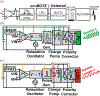
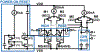
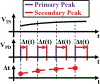
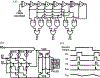
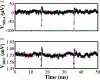
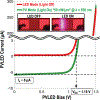
Recording neural activity in live animals in vivo with minimal tissue damage is one of the major barriers to understanding the nervous system. This paper presents the technology for a tetherless opto-electronic neural interface based on 180 nm CMOS circuits, heterogeneously integrated with an AlGaAs diode that functions as both a photovoltaic and light emitting diode. These microscale opto-electrically transduced electrodes (MOTEs) are powered by and communicate through an optical interface, simultaneously enabling high temporal-resolution electrical measurements without a tether or a bulky RF coil. The MOTE presented here is 250 μm × 57 μm, consumes 1 μW of electrical power, and is capable of capturing and encoding neural signals before transmitting the encoded signals. The measured noise floor is as low as 15 μVRMS at a 15 kHz bandwidth.
Figures
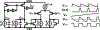


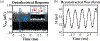
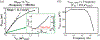
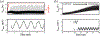


References
-
- Schenck JF, “The role of magnetic susceptibility in magnetic resonance imaging: MRI magnetic compatibility of the first and second kinds,” Medical Physics, vol. 23, no. 6, pp. 815–850, June 1996. - PubMed
-
- “https://www.braininitiative.nih.gov/,” [Online].
-
- Johnson B, Peace ST, Wang A, Cleland TA and Molnar A, “A 768-Channel CMOS Microelectrode Array With Angle Sensitive Pixels for Neuronal Recording,” in lEEESensorsJournal, vol. 13, no. 9, pp. 3211–3218, September 2013.
-
- Dragas J, Viswam V, Shadmani A, Chen Y, Bounik R, Stettler A, Radivojevic M, Geissler S, Obien MEJ, Müller J and Hierlemann A, “In Vitro Multi-Functional Microelectrode Array Featuring 59 760 Electrodes, 2048 Electrophysiology Channels, Stimulation, Impedance Measurement, and Neurotransmitter Detection Channels,” in IEEE Journal of Solid-State Circuits, vol. 52, no. 6, pp. 1576–1590, June 2017. - PMC - PubMed

