Enhanced Dendritic Compartmentalization in Human Cortical Neurons
- PMID: 30340039
- PMCID: PMC6197488
- DOI: 10.1016/j.cell.2018.08.045
Enhanced Dendritic Compartmentalization in Human Cortical Neurons
Abstract
The biophysical features of neurons shape information processing in the brain. Cortical neurons are larger in humans than in other species, but it is unclear how their size affects synaptic integration. Here, we perform direct electrical recordings from human dendrites and report enhanced electrical compartmentalization in layer 5 pyramidal neurons. Compared to rat dendrites, distal human dendrites provide limited excitation to the soma, even in the presence of dendritic spikes. Human somas also exhibit less bursting due to reduced recruitment of dendritic electrogenesis. Finally, we find that decreased ion channel densities result in higher input resistance and underlie the lower coupling of human dendrites. We conclude that the increased length of human neurons alters their input-output properties, which will impact cortical computation. VIDEO ABSTRACT.
Keywords: biophysics; compartmentalization; computation; cortex; dendrite; human; ion channels; neuron; patch-clamp.
Published by Elsevier Inc.
Figures
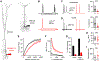


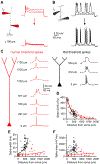

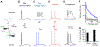

Comment in
-
Human Cortical Dendrites: Stretched to Perform Better?Cell. 2018 Oct 18;175(3):635-637. doi: 10.1016/j.cell.2018.09.052. Cell. 2018. PMID: 30340035
-
Are Human Dendrites Different?Trends Cogn Sci. 2020 Jun;24(6):411-412. doi: 10.1016/j.tics.2020.03.002. Epub 2020 Mar 30. Trends Cogn Sci. 2020. PMID: 32392467 Free PMC article.
References
-
- Beaulieu-Laroche L, and Harnett MT (2018). Dendritic Spines Prevent Synaptic Voltage Clamp. Neuron 97, 75–82.e73. - PubMed
-
- Carnevale NT, and Hines ML (2006). The NEURON Book (Cambridge: Cambridge University Press; ).
-
- Connors BW, Gutnick MJ, and Prince DA (1982). Electrophysiological properties of neocortical neurons in vitro. Journal of neurophysiology 48, 1302–1320. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

