The Shrinking Brain: Cerebral Atrophy Following Traumatic Brain Injury
- PMID: 30341741
- PMCID: PMC6757025
- DOI: 10.1007/s10439-018-02148-2
The Shrinking Brain: Cerebral Atrophy Following Traumatic Brain Injury
Abstract
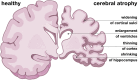
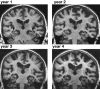
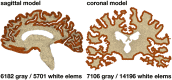
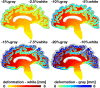
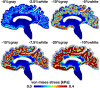
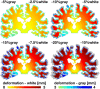
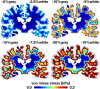
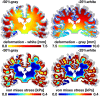
Cerebral atrophy in response to traumatic brain injury is a well-documented phenomenon in both primary investigations and review articles. Recent atrophy studies focus on exploring the region-specific patterns of cerebral atrophy; yet, there is no study that analyzes and synthesizes the emerging atrophy patterns in a single comprehensive review. Here we attempt to fill this gap in our current knowledge by integrating the current literature into a cohesive theory of preferential brain tissue loss and by identifying common risk factors for accelerated atrophy progression. Our review reveals that observations for mild traumatic brain injury remain inconclusive, whereas observations for moderate-to-severe traumatic brain injury converge towards robust patterns: brain tissue loss is on the order of 5% per year, and occurs in the form of generalized atrophy, across the entire brain, or focal atrophy, in specific brain regions. The most common regions of focal atrophy are the thalamus, hippocampus, and cerebellum in gray matter and the corpus callosum, corona radiata, and brainstem in white matter. We illustrate the differences of generalized and focal gray and white matter atrophy on emerging deformation and stress profiles across the whole brain using computational simulation. The characteristic features of our atrophy simulations-a widening of the cortical sulci, a gradual enlargement of the ventricles, and a pronounced cortical thinning-agree well with clinical observations. Understanding region-specific atrophy patterns in response to traumatic brain injury has significant implications in modeling, simulating, and predicting injury outcomes. Computational modeling of brain atrophy could open new strategies for physicians to make informed decisions for whom, how, and when to administer pharmaceutical treatment to manage the chronic loss of brain structure and function.
Keywords: Cerebral atrophy; Computational simulation; Finite element modeling; Neurodegeneration; Traumatic brain injury.
Figures
References
-
- Bowen DM, Allen SJ, Benton JS, Goodhardt MJ, Haan EA, Palmer AM, Sims NR, Smith CCT, Spillane JA, Esiri MM, Neary D, Snowdon JS, Wilcock GK, Davison AN. Biochemical assessment of serotonergic and cholinergic dysfunction and cerebral atrophy in Alzheimers-disease. J. Neurochem. 1983;41(1):266–272. doi: 10.1111/j.1471-4159.1983.tb11838.x. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

