A pooled analysis of individual patient data from National Clinical Trials Network clinical trials of concurrent chemoradiotherapy for limited-stage small cell lung cancer in elderly patients versus younger patients
- PMID: 30343497
- PMCID: PMC6340765
- DOI: 10.1002/cncr.31813
A pooled analysis of individual patient data from National Clinical Trials Network clinical trials of concurrent chemoradiotherapy for limited-stage small cell lung cancer in elderly patients versus younger patients
Abstract
Background: Platinum and etoposide with thoracic radiation followed by prophylactic cranial irradiation constitute the standard treatment for limited-stage small cell lung cancer (LS-SCLC). Many patients with LS-SCLC are elderly with comorbidities.
Methods: Individual patient data were collected from 11 phase 2 or 3 trials for LS-SCLC conducted by the National Clinical Trials Network and activated from 1990 to 2010. The primary endpoint was overall survival (OS); the secondary endpoints were progression-free survival (PFS), the rate of severe adverse events, and off-treatment reasons. The outcomes were compared for patients 70 years old or older (elderly patients) and patients younger than 70 years (younger patients).
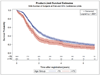
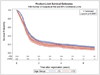
Results: Individual patient data from 1049 younger patients (81%) and 254 elderly patients (19%) were analyzed. In the multivariate model, elderly patients, in comparison with younger patients, had worse OS (hazard ratio [HR], 1.38; 95% confidence interval [CI], 1.18-1.63; median OS for elderly patients, 17.8 months; OS for younger patients, 23.5 months) and worse PFS (HR, 1.19; 95% CI, 1.03-1.39; median PFS for elderly patients, 10.6 months; median PFS for younger patients, 12.3 months). Elderly patients, in comparison with younger patients, experienced more grade 5 adverse events (8% vs 3%; P < .01) and more grade 3 or higher dyspnea (11% vs 7%; P = .03) but less grade 3 or higher esophagitis/dysphagia (14% vs 19%; P = .04) and less grade 3 or higher vomiting (11% vs 17%; P = .01). Elderly patients completed treatment less often, discontinued treatment because of adverse events and patient refusal more frequently, and died during treatment more frequently.
Conclusions: Elderly patients with LS-SCLC have worse PFS and OS and more difficulty in tolerating therapy. Future trials should incorporate assessments of elderly patients, novel monitoring of adverse events, and more tolerable radiation and systemic therapies.
Keywords: adverse events related to age; chemotherapy; clinical trial; small cell lung cancer; thoracic radiation therapy.
© 2018 American Cancer Society.
Conflict of interest statement
Conflict of interest:
The following authors do not report any conflicts of interest: Stinchcombe, Cohen, Pang, Le, Fan, Bogart, Vokes, Horn, Edelman, Komaki, Schild, Bogart, Thomas, and Wang
Dr. Ganti reports grants and personal fees from Pfizer, grants from New Link Genetics, personal fees from Ariad Pharmaceuticals, grants from Amgen, grants from AstraZeneca, grants from Merck, grants from Janssen, grants from Bristol-Myers Squibb, personal fees from Biodesix, outside the submitted work.
Figures
References
-
- National Cancer Institute: SEER stat fact sheets: Lung and bronchus cancer. https://seer.cancer.gov/statfacts/html/lungb.html-accesse 3/19/2018.
-
- Govindan R, Page N, Morgensztern D, et al. Changing epidemiology of small-cell lung cancer in the United States over the last 30 years: analysis of the surveillance, epidemiologic, and end results database. J Clin Oncol 2006;24: 4539–4544. - PubMed
-
- Fruh M, De Ruysscher D, Popat S, et al. Small-cell lung cancer (SCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2013;24 Suppl 6: vi99–105. - PubMed
-
- Sun JM, Ahn YC, Choi EK, et al. Phase III trial of concurrent thoracic radiotherapy with either first- or third-cycle chemotherapy for limited-disease small-cell lung cancer (correction). Annals of Oncology. 2014;25: 1672. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical