Antibacterial and Bioactive Surface Modifications of Titanium Implants by PCL/TiO₂ Nanocomposite Coatings
- PMID: 30347811
- PMCID: PMC6215281
- DOI: 10.3390/nano8100860
Antibacterial and Bioactive Surface Modifications of Titanium Implants by PCL/TiO₂ Nanocomposite Coatings
Abstract
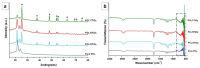
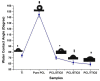
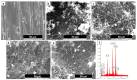
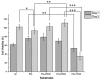
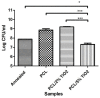
Surface modification of biomedical implants is an established strategy to improve tissue regeneration, osseointegration and also to minimize the bacterial accumulation. In the present study, electrospun poly(ε-caprolactone)/titania (PCL/TiO₂) nanocomposite coatings were developed on commercially pure titanium (cpTi) substrates for an improved biological and antibacterial properties for bone tissue engineering. TiO₂ nanoparticles in various amounts (2, 5, and 7 wt %) were incorporated into a biodegradable PCL matrix to form a homogeneous solution. Further, PCL/TiO₂ coatings on cpTi were obtained by electrospinning of PCL/TiO₂ solution onto the substrate. The resulted coatings were structurally characterized and inspected by employing scanning electron microscope (SEM), X-ray diffraction (XRD), and Fourier transform infrared (FTIR) spectroscopy. Given the potential biological applications of PCL/TiO₂ coated cpTi substrates, the apatite-forming capacity was examined by immersing in simulated body fluid (SBF) for upto 21 days. Biocompatibility has been evaluated through adhesion/proliferation of hFOB osteoblast cell lines and cytotoxicity by MTT assay. Antimicrobial activity of PCL/TiO₂ nanocomposites has been tested using UV light against gram-positive Staphylococcus aureus (S.aureus). The resulting surface displays good bioactive properties against osteoblast cell lines with increased viability of 40% at day 3 and superior antibacterial property against S.aureus with a significant reduction of bacteria to almost 76%. Surface modification by PCL/TiO₂ nanocomposites makes a viable approach for improving dual properties, i.e., biological and antibacterial properties on titanium implants which might be used to prevent implant-associated infections and promoting cell attachment of orthopedic devices at the same time.
Keywords: TiO2 photocatalytic; antibacterial coatings; electrospinning; nanocomposite coatings; orthopedic infections; titanium.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Electrospun Polycaprolactone Fibrous Membranes Containing Ag, TiO₂ and Na₂Ti₆O13 Particles for Potential Use in Bone Regeneration.Membranes (Basel). 2019 Jan 10;9(1):12. doi: 10.3390/membranes9010012. Membranes (Basel). 2019. PMID: 30634630 Free PMC article.
-
Microstructure and cytotoxicity evaluation of duplex-treated silver-containing antibacterial TiO₂ coatings.Mater Sci Eng C Mater Biol Appl. 2014 Dec;45:402-10. doi: 10.1016/j.msec.2014.07.002. Epub 2014 Sep 19. Mater Sci Eng C Mater Biol Appl. 2014. PMID: 25491845
-
Hydroxyapatite-TiO(2)-based nanocomposites synthesized in supercritical CO(2) for bone tissue engineering: physical and mechanical properties.ACS Appl Mater Interfaces. 2014 Oct 8;6(19):16918-31. doi: 10.1021/am5044888. Epub 2014 Sep 23. ACS Appl Mater Interfaces. 2014. PMID: 25184699
-
Review of titanium surface modification techniques and coatings for antibacterial applications.Acta Biomater. 2019 Jan 1;83:37-54. doi: 10.1016/j.actbio.2018.10.036. Epub 2018 Oct 26. Acta Biomater. 2019. PMID: 30541702 Review.
-
Antibacterial Coatings for Titanium Implants: Recent Trends and Future Perspectives.Antibiotics (Basel). 2022 Nov 29;11(12):1719. doi: 10.3390/antibiotics11121719. Antibiotics (Basel). 2022. PMID: 36551376 Free PMC article. Review.
Cited by
-
Titanium(IV) Oxo-Complex with Acetylsalicylic Acid Ligand and Its Polymer Composites: Synthesis, Structure, Spectroscopic Characterization, and Photocatalytic Activity.Materials (Basel). 2022 Jun 22;15(13):4408. doi: 10.3390/ma15134408. Materials (Basel). 2022. PMID: 35806533 Free PMC article.
-
Engineered titania nanomaterials in advanced clinical applications.Beilstein J Nanotechnol. 2022 Feb 14;13:201-218. doi: 10.3762/bjnano.13.15. eCollection 2022. Beilstein J Nanotechnol. 2022. PMID: 35223351 Free PMC article. Review.
-
A Comprehensive Review on Bio-Nanomaterials for Medical Implants and Feasibility Studies on Fabrication of Such Implants by Additive Manufacturing Technique.Materials (Basel). 2019 Dec 23;13(1):92. doi: 10.3390/ma13010092. Materials (Basel). 2019. PMID: 31878040 Free PMC article. Review.
-
Recent Advancements in Materials and Coatings for Biomedical Implants.Gels. 2022 May 21;8(5):323. doi: 10.3390/gels8050323. Gels. 2022. PMID: 35621621 Free PMC article. Review.
-
Enhanced healing of critical-sized bone defects using degradable scaffolds with tailored composition through immunomodulation and angiogenesis.Bioact Mater. 2024 Oct 28;44:371-388. doi: 10.1016/j.bioactmat.2024.10.018. eCollection 2025 Feb. Bioact Mater. 2024. PMID: 39539516 Free PMC article.
References
-
- Douglas Scott II R. The Direct Medical Costs of Healthcare-Associated Infections in U.S. Hospitals and the Benefits of Prevention. Centers for Disease Control and Prevention; Atlanta, GA, USA: 2009.
LinkOut - more resources
Full Text Sources

