Disease- and treatment-associated acquired glucocorticoid resistance
- PMID: 30352419
- PMCID: PMC6280593
- DOI: 10.1530/EC-18-0421
Disease- and treatment-associated acquired glucocorticoid resistance
Abstract
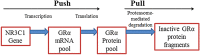
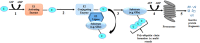
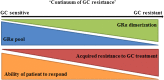
The development of resistance to glucocorticoids (GCs) in therapeutic regimens poses a major threat. Generally, GC resistance is congenital or acquired over time as a result of disease progression, prolonged GC treatment or, in some cases, both. Essentially, disruptions in the function and/or pool of the glucocorticoid receptor α (GRα) underlie this resistance. Many studies have detailed how alterations in GRα function lead to diminished GC sensitivity; however, the current review highlights the wealth of data concerning reductions in the GRα pool, mediated by disease-associated and treatment-associated effects, which contribute to a significant decrease in GC sensitivity. Additionally, the current understanding of the molecular mechanisms involved in driving reductions in the GRα pool is discussed. After highlighting the importance of maintaining the level of the GRα pool to combat GC resistance, we present current strategies and argue that future strategies to prevent GC resistance should involve biased ligands with a predisposition for reduced GR dimerization, a strategy originally proposed as the SEMOGRAM-SEDIGRAM concept to reduce the side-effect profile of GCs.
Keywords: GRα downregulation; acquired resistance; biased ligands; glucocorticoid receptor; glucocorticoid resistance.
Figures

Similar articles
-
GR Dimerization and the Impact of GR Dimerization on GR Protein Stability and Half-Life.Front Immunol. 2019 Jul 17;10:1693. doi: 10.3389/fimmu.2019.01693. eCollection 2019. Front Immunol. 2019. PMID: 31379877 Free PMC article. Review.
-
Acquired Glucocorticoid Resistance Due to Homologous Glucocorticoid Receptor Downregulation: A Modern Look at an Age-Old Problem.Cells. 2021 Sep 24;10(10):2529. doi: 10.3390/cells10102529. Cells. 2021. PMID: 34685511 Free PMC article. Review.
-
Relative contribution of molecular mechanisms to cumulative ligand-mediated downregulation of GRα.Biochem Biophys Res Commun. 2022 Apr 30;602:113-119. doi: 10.1016/j.bbrc.2022.02.102. Epub 2022 Feb 25. Biochem Biophys Res Commun. 2022. PMID: 35263658
-
Expression of glucocorticoid receptor isoforms and associations with serine/arginine-rich protein 30c and 40 in patients with systemic lupus erythematosus.Clin Exp Rheumatol. 2015 Mar-Apr;33(2):225-33. Epub 2015 Feb 9. Clin Exp Rheumatol. 2015. PMID: 25665148
-
Novel role for receptor dimerization in post-translational processing and turnover of the GRα.Sci Rep. 2018 Sep 24;8(1):14266. doi: 10.1038/s41598-018-32440-z. Sci Rep. 2018. PMID: 30250038 Free PMC article.
Cited by
-
Dimerization of the Glucocorticoid Receptor and Its Importance in (Patho)physiology: A Primer.Cells. 2022 Feb 15;11(4):683. doi: 10.3390/cells11040683. Cells. 2022. PMID: 35203332 Free PMC article. Review.
-
GR Dimerization and the Impact of GR Dimerization on GR Protein Stability and Half-Life.Front Immunol. 2019 Jul 17;10:1693. doi: 10.3389/fimmu.2019.01693. eCollection 2019. Front Immunol. 2019. PMID: 31379877 Free PMC article. Review.
-
Glucocorticoid Resistance: Interference between the Glucocorticoid Receptor and the MAPK Signalling Pathways.Int J Mol Sci. 2021 Sep 17;22(18):10049. doi: 10.3390/ijms221810049. Int J Mol Sci. 2021. PMID: 34576214 Free PMC article. Review.
-
Pharmacokinetics and Pharmacodynamics of Repository Corticotropin Injection Compared With Synthetic ACTH1-24 Depot and Methylprednisolone in Healthy Subjects.Clin Pharmacol Drug Dev. 2022 Apr;11(4):502-515. doi: 10.1002/cpdd.1020. Epub 2021 Sep 15. Clin Pharmacol Drug Dev. 2022. PMID: 34528408 Free PMC article. Clinical Trial.
-
Association between glucocorticoid receptor beta and steroid resistance: A systematic review.Immun Inflamm Dis. 2024 Jan;12(1):e1137. doi: 10.1002/iid3.1137. Immun Inflamm Dis. 2024. PMID: 38270313 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

