Doxapram Treatment and Diaphragmatic Activity in Preterm Infants
- PMID: 30352445
- PMCID: PMC6425813
- DOI: 10.1159/000493359
Doxapram Treatment and Diaphragmatic Activity in Preterm Infants
Abstract
Background: Doxapram is a treatment option for severe apnea of prematurity (AOP). However, the effect of doxapram on the diaphragm, the main respiratory muscle, is not known.
Objectives: To investigate the effect of doxapram on diaphragmatic activity measured with transcutaneous electromyography of the diaphragm (dEMG).
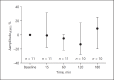
Methods: A pilot study was conducted in a tertiary neonatal intensive care unit. Diaphragmatic activity was measured from 30 min before up to 3 h after the start of doxapram treatment. dEMG parameters were compared to baseline (5 min before doxapram treatment) and at 15, 60, 120 and 180 min after the start of doxapram infusion.
Results: Eleven preterm infants were included with a mean gestational age of 25.5 ± 1.2 weeks and birth weight of 831 ± 129 g. The amplitudedEMG, peakdEMG and tonicdEMG values did not change in the 3 h after the start of doxapram infusion compared to baseline. Clinically, the number of apnea episodes in the 24 h after doxapram treatment decreased significantly.
Conclusion: Doxapram infusion does not alter diaphragmatic activity measured with transcutaneous dEMG in preterm infants with AOP, indicating that its working mechanism is primarily on respiratory drive and not on respiratory muscle activity.
Keywords: Diaphragm; Doxapram; Transcutaneous electromyography.
© 2018 The Author(s) Published by S. Karger AG, Basel.
Figures
References
-
- Eichenwald EC, Committee on Fetus and Newborn, American Academy of Pediatrics Apnea of Prematurity. Pediatrics. 2016 Jan;137((1)):e20153757. - PubMed
-
- Hascoet JM, Hamon I, Boutroy MJ. Risks and benefits of therapies for apnoea in premature infants. Drug Saf. 2000 Nov;23((5)):363–79. - PubMed
-
- Flint R, Halbmeijer N, Meesters N, van Rosmalen J, Reiss I, van Dijk M, et al. Retrospective study shows that doxapram therapy avoided the need for endotracheal intubation in most premature neonates. Acta Paediatr. 2017 May;106((5)):733–9. - PubMed
-
- Barrington KJ, Finer NN, Peters KL, Barton J. Physiologic effects of doxapram in idiopathic apnea of prematurity. J Pediatr. 1986 Jan;108((1)):124–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical