Usefulness of myeloperoxidase as a biomarker for the ranking of pulmonary toxicity of nanomaterials
- PMID: 30352603
- PMCID: PMC6199695
- DOI: 10.1186/s12989-018-0277-x
Usefulness of myeloperoxidase as a biomarker for the ranking of pulmonary toxicity of nanomaterials
Abstract
Background: In order to examine whether myeloperoxidase (MPO) can be a useful marker for evaluating the pulmonary toxicity of nanomaterials, we analyzed MPO protein in bronchoalveolar lavage fluid (BALF) samples obtained from previous examinations of a rat model. In those examinations we performed intratracheal instillation exposures (dose: 0.2-1.0 mg) and inhalation exposures (exposure concentration: 0.32-10.4 mg/m3) using 9 and 4 nanomaterials with different toxicities, respectively. Based on those previous studies, we set Nickel oxide nanoparticles (NiO), cerium dioxide nanoparticles (CeO2), multi wall carbon nanotubes with short or long length (MWCNT (S) and MWCNT (L)), and single wall carbon nanotube (SWCNT) as chemicals with high toxicity; and titanium dioxide nanoparticles (TiO2 (P90) and TiO2 (Rutile)), zinc oxide nanoparticles (ZnO), and toner with external additives including nanoparticles as chemicals with low toxicity. We measured the concentration of MPO in BALF samples from rats from 3 days to 6 months following a single intratracheal instillation, and from 3 days to 3 months after the end of inhalation exposure.
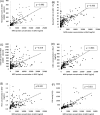
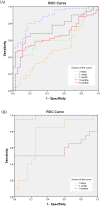
Results: Intratracheal instillation of high toxicity NiO, CeO2, MWCNT (S), MWCNT (L), and SWCNT persistently increased the concentration of MPO, and inhalation of NiO and CeO2 increased the MPO in BALF. By contrast, intratracheal instillation of low toxicity TiO2 (P90), TiO2 (Rutile), ZnO, and toner increased the concentration of MPO in BALF only transiently, and inhalation of TiO2 (Rutile) and ZnO induced almost no increase of the MPO. The concentration of MPO correlated with the number of total cells and neutrophils, the concentration of chemokines for neutrophils (cytokine-induced neutrophil chemoattractant (CINC)-1 and heme oxygenase (HO)-1), and the activity of released lactate dehydrogenase (LDH) in BALF. The results from the receiver operating characteristics (ROC) for the toxicity of chemicals by the concentration of MPO proteins in the intratracheal instillation and inhalation exposures showed that the largest areas under the curves (AUC) s in both examinations occurred at 1 month after exposure.
Conclusion: These data suggest that MPO can be a useful biomarker for the ranking of the pulmonary toxicity of nanomaterials, especially at 1 month after exposure, in both intratracheal instillation and inhalation exposure.
Keywords: Biomarker; Inhalation; Intratracheal instillation; Myeloperoxidase; Nanomaterial; Pulmonary toxicity; Rat.
Conflict of interest statement
Ethics approval and consent to participate
All procedures and animal handling were done according to the guidelines described in the Japanese Guide for the Care and Use of Laboratory Animals as approved by the Animal Care and Use Committee, University of Occupational and Environmental Health, Japan.
Consent for publication
Not required as no human data presented.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


Similar articles
-
Comparison of pulmonary inflammatory responses following intratracheal instillation and inhalation of nanoparticles.Nanotoxicology. 2016;10(5):607-18. doi: 10.3109/17435390.2015.1104740. Epub 2015 Nov 11. Nanotoxicology. 2016. PMID: 26558952
-
Assessment of Cytokine-Induced Neutrophil Chemoattractants as Biomarkers for Prediction of Pulmonary Toxicity of Nanomaterials.Nanomaterials (Basel). 2020 Aug 9;10(8):1563. doi: 10.3390/nano10081563. Nanomaterials (Basel). 2020. PMID: 32784876 Free PMC article.
-
Evaluation of Pulmonary Toxicity of Zinc Oxide Nanoparticles Following Inhalation and Intratracheal Instillation.Int J Mol Sci. 2016 Aug 1;17(8):1241. doi: 10.3390/ijms17081241. Int J Mol Sci. 2016. PMID: 27490535 Free PMC article.
-
Inhalation toxicity assessment of carbon-based nanoparticles.Acc Chem Res. 2013 Mar 19;46(3):770-81. doi: 10.1021/ar200311b. Epub 2012 May 11. Acc Chem Res. 2013. PMID: 22574947 Review.
-
Usefulness of Intratracheal Instillation Studies for Estimating Nanoparticle-Induced Pulmonary Toxicity.Int J Mol Sci. 2016 Jan 27;17(2):165. doi: 10.3390/ijms17020165. Int J Mol Sci. 2016. PMID: 26828483 Free PMC article. Review.
Cited by
-
In Vivo Comparative Study on Acute and Sub-acute Biological Effects Induced by Ultrafine Particles of Different Anthropogenic Sources in BALB/c Mice.Int J Mol Sci. 2019 Jun 8;20(11):2805. doi: 10.3390/ijms20112805. Int J Mol Sci. 2019. PMID: 31181746 Free PMC article.
-
Silica nanoparticles induce lung inflammation in mice via ROS/PARP/TRPM2 signaling-mediated lysosome impairment and autophagy dysfunction.Part Fibre Toxicol. 2020 Jun 8;17(1):23. doi: 10.1186/s12989-020-00353-3. Part Fibre Toxicol. 2020. PMID: 32513195 Free PMC article.
-
Examination of Surfactant Protein D as a Biomarker for Evaluating Pulmonary Toxicity of Nanomaterials in Rat.Int J Mol Sci. 2021 Apr 28;22(9):4635. doi: 10.3390/ijms22094635. Int J Mol Sci. 2021. PMID: 33924924 Free PMC article.
-
Long-term observation of pulmonary toxicity of toner with external additives following a single intratracheal instillation in rats.J Occup Health. 2020 Jan;62(1):e12146. doi: 10.1002/1348-9585.12146. J Occup Health. 2020. PMID: 32710690 Free PMC article.
-
The pulmonary effects of nickel-containing nanoparticles: Cytotoxicity, genotoxicity, carcinogenicity, and their underlying mechanisms.Environ Sci Nano. 2024 May 1;11(5):1817-1846. doi: 10.1039/d3en00929g. Epub 2024 Mar 21. Environ Sci Nano. 2024. PMID: 38984270 Free PMC article.
References
-
- ISO N . Terminology and definitions for Nano-objects – nanoparticle, nanofibre and Nanoplate. 2008.
-
- Oberdörster G, Maynard A, Donaldson K, Castranova V, Fitzpatrick J, Ausman K, et al. Principles for characterizing the potential human health effects from exposure to nanomaterials: elements of a screening strategy. Particle and Fibre Toxicology. 2005;2:8. doi: 10.1186/1743-8977-2-8. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

