A Type 1 Diabetes Genetic Risk Score Can Identify Patients With GAD65 Autoantibody-Positive Type 2 Diabetes Who Rapidly Progress to Insulin Therapy
- PMID: 30352895
- PMCID: PMC6828553
- DOI: 10.2337/dc18-0431
A Type 1 Diabetes Genetic Risk Score Can Identify Patients With GAD65 Autoantibody-Positive Type 2 Diabetes Who Rapidly Progress to Insulin Therapy
Abstract
Objective: Progression to insulin therapy in clinically diagnosed type 2 diabetes is highly variable. GAD65 autoantibodies (GADA) are associated with faster progression, but their predictive value is limited. We aimed to determine if a type 1 diabetes genetic risk score (T1D GRS) could predict rapid progression to insulin treatment over and above GADA testing.
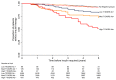
Research design and methods: We examined the relationship between T1D GRS, GADA (negative or positive), and rapid insulin requirement (within 5 years) using Kaplan-Meier survival analysis and Cox regression in 8,608 participants with clinical type 2 diabetes (onset >35 years and treated without insulin for ≥6 months). T1D GRS was both analyzed continuously (as standardized scores) and categorized based on previously reported centiles of a population with type 1 diabetes (<5th [low], 5th-50th [medium], and >50th [high]).
Results: In GADA-positive participants (3.3%), those with higher T1D GRS progressed to insulin more quickly: probability of insulin requirement at 5 years (95% CI): 47.9% (35.0%, 62.78%) (high T1D GRS) vs. 27.6% (20.5%, 36.5%) (medium T1D GRS) vs. 17.6% (11.2%, 27.2%) (low T1D GRS); P = 0.001. In contrast, T1D GRS did not predict rapid insulin requirement in GADA-negative participants (P = 0.4). In Cox regression analysis with adjustment for age of diagnosis, BMI, and cohort, T1D GRS was independently associated with time to insulin only in the presence of GADA: hazard ratio per SD increase was 1.48 (1.15, 1.90); P = 0.002.
Conclusions: A T1D GRS alters the clinical implications of a positive GADA test in patients with clinical type 2 diabetes and is independent of and additive to clinical features.
© 2018 by the American Diabetes Association.
Conflict of interest statement
No potential conflicts of interest relevant to this article were reported.
Figures
References
-
- U.K. Prospective Diabetes Study Group: U.K. Prospective Diabetes Study 16: Overview of 6 Years' therapy of Type II Diabetes: A Progressive Disease. Diabetes. 1995;44:1249. - PubMed
-
- Groop LC, Bottazzo GF, Doniach D. Islet Cell Antibodies Identify Latent Type I Diabetes in Patients Aged 35–75 Years at Diagnosis. Diabetes. 1986;35:237. - PubMed
-
- Turner R, Stratton I, Horton V, Manley S, Zimmet P, Mackay IR, Shattock M, Bottazzo GF, Holman R. UKPDS 25: autoantibodies to islet-cell cytoplasm and glutamic acid decarboxylase for prediction of insulin requirement in type 2 diabetes. The Lancet. 350:1288–1293. - PubMed
-
- Tuomi T, Groop LC, Zimmet PZ, Rowley MJ, Knowles W, Mackay IR. Antibodies to Glutamic Acid Decarboxylase Reveal Latent Autoimmune Diabetes Mellitus in Adults With a Non—Insulin-Dependent Onset of Disease. Diabetes. 1993;42:359. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

