Progressive waves of IL-1β release by primary human monocytes via sequential activation of vesicular and gasdermin D-mediated secretory pathways
- PMID: 30352992
- PMCID: PMC6199333
- DOI: 10.1038/s41419-018-1121-9
Progressive waves of IL-1β release by primary human monocytes via sequential activation of vesicular and gasdermin D-mediated secretory pathways
Abstract
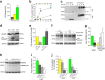
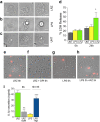
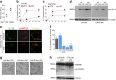
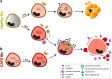
IL-1β is an essential cytokine, but its release needs to be strictly controlled to avoid severe inflammatory manifestations. Lacking a signal sequence, IL-1β does not follow the endoplasmic reticulum-Golgi route. Several pathways have been proposed to mediate its release. One involves the translocation of pro-IL-1β into intracellular vesicles of lysosomal origin that eventually fuse with the plasma membrane. Another exploits pores formed on the plasma membrane upon proteolytic cleavage of gasdermin D (GSDMD). Here we investigated how primary monocytes-the main source of IL-1β in humans-control IL-1β release in response to pro-inflammatory stimuli of increasing intensity and found that two different routes are induced depending on the strength of activation. Triggering of Toll-like receptor 4 (TLR4) by LPS induces slow IL-1β release through LAMP2A+ vesicles. In contrast, the simultaneous stimulation of TLR2, TLR4 and TLR7/8 drives high levels of ROS, GSDMD cleavage and faster IL-1β secretion. Drugs blocking ROS production prevent GSDMD cleavage supporting a role of oxidative stress in GSDMD-mediated secretion. Singly stimulated monocytes undergo apoptosis, whereas triple stimulation triggers pyroptosis, which might amplify inflammation. In both cases, however, IL-1β secretion precedes cell death. Inhibition of caspases 4/5 prevents GSDMD cleavage and pore-mediated secretion, but not vesicular release. The two pathways also display other distinct pharmacologic sensitivities that reflect the underlying mechanisms. Remarkably, single TLR4 stimulation is sufficient to activate massive, GSDMD-mediated IL-1β secretion in monocytes from patients affected by Cryopyrin Associated Periodic Syndrome (CAPS), an autoinflammatory disease linked to NLRP3 mutations. The exaggerated sensitivity to activation correlates with high basal ROS levels in CAPS monocytes. In conclusion, the vesicular pathway limits IL-1β release upon low pathogen load while stronger stimulation or concomitant cell stress induce instead uncontrolled secretion via GSDMD leading to detrimental inflammatory manifestations.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



Similar articles
-
Multiple TLRs elicit alternative NLRP3 inflammasome activation in primary human monocytes independent of RIPK1 kinase activity.Front Immunol. 2023 Oct 26;14:1092799. doi: 10.3389/fimmu.2023.1092799. eCollection 2023. Front Immunol. 2023. PMID: 37954581 Free PMC article.
-
Toxoplasma gondii activates a Syk-CARD9-NF-κB signaling axis and gasdermin D-independent release of IL-1β during infection of primary human monocytes.PLoS Pathog. 2019 Aug 26;15(8):e1007923. doi: 10.1371/journal.ppat.1007923. eCollection 2019 Aug. PLoS Pathog. 2019. PMID: 31449558 Free PMC article.
-
Interleukin-1β Maturation Triggers Its Relocation to the Plasma Membrane for Gasdermin-D-Dependent and -Independent Secretion.Cell Rep. 2018 Aug 7;24(6):1425-1433. doi: 10.1016/j.celrep.2018.07.027. Cell Rep. 2018. PMID: 30089254
-
Uncoupled pyroptosis and IL-1β secretion downstream of inflammasome signaling.Front Immunol. 2023 Apr 6;14:1128358. doi: 10.3389/fimmu.2023.1128358. eCollection 2023. Front Immunol. 2023. PMID: 37090724 Free PMC article. Review.
-
Emerging insights into molecular mechanisms underlying pyroptosis and functions of inflammasomes in diseases.J Cell Physiol. 2020 Apr;235(4):3207-3221. doi: 10.1002/jcp.29268. Epub 2019 Oct 17. J Cell Physiol. 2020. PMID: 31621910 Review.
Cited by
-
Gasdermin D Protects from Melioidosis through Pyroptosis and Direct Killing of Bacteria.J Immunol. 2019 Jun 15;202(12):3468-3473. doi: 10.4049/jimmunol.1900045. Epub 2019 Apr 29. J Immunol. 2019. PMID: 31036765 Free PMC article.
-
Post-Transcriptional Control of mRNA Metabolism and Protein Secretion: The Third Level of Regulation within the NF-κB System.Biomedicines. 2022 Aug 29;10(9):2108. doi: 10.3390/biomedicines10092108. Biomedicines. 2022. PMID: 36140209 Free PMC article. Review.
-
Role of signal transduction pathways in IL-1β-induced apoptosis: Pathological and therapeutic aspects.Immun Inflamm Dis. 2023 Jan;11(1):e762. doi: 10.1002/iid3.762. Immun Inflamm Dis. 2023. PMID: 36705417 Free PMC article. Review.
-
Herbal melanin induces interleukin-1β secretion and production by human THP-1 monocytes via Toll-like receptor 2 and p38 MAPK activation.Exp Ther Med. 2021 Oct;22(4):1081. doi: 10.3892/etm.2021.10515. Epub 2021 Jul 29. Exp Ther Med. 2021. PMID: 34447474 Free PMC article.
-
NLRP3 inflammasome triggers interleukin-37 release from human monocytes.Eur J Immunol. 2022 Jul;52(7):1141-1157. doi: 10.1002/eji.202149724. Epub 2022 Apr 28. Eur J Immunol. 2022. PMID: 35429346 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

