Irradiance controls photodynamic efficacy and tissue heating in experimental tumours: implication for interstitial PDT of locally advanced cancer
- PMID: 30353043
- PMCID: PMC6251027
- DOI: 10.1038/s41416-018-0210-y
Irradiance controls photodynamic efficacy and tissue heating in experimental tumours: implication for interstitial PDT of locally advanced cancer
Abstract
Background: Currently delivered light dose (J/cm2) is the principal parameter guiding interstitial photodynamic therapy (I-PDT) of refractory locally advanced cancer. The aim of this study was to investigate the impact of light dose rate (irradiance, mW/cm2) and associated heating on tumour response and cure.
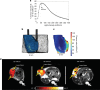
Methods: Finite-element modeling was used to compute intratumoural irradiance and dose to guide Photofrin® I-PDT in locally advanced SCCVII in C3H mice and large VX2 neck tumours in New Zealand White rabbits. Light-induced tissue heating in mice was studied with real-time magnetic resonance thermometry.
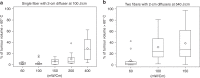
Results: In the mouse model, cure rates of 70-90% were obtained with I-PDT using 8.4-245 mW/cm2 and ≥45 J/cm2 in 100% of the SCCVII tumour. Increasing irradiance was associated with increase in tissue heating. I-PDT with Photofrin® resulted in significantly (p < 0.05) higher cure rate compared to light delivery alone at same irradiance and light dose. Local control and/or cures of VX2 were obtained using I-PDT with 16.5-398 mW/cm2 and ≥45 J/cm2 in 100% of the tumour.
Conclusion: In Photofrin®-mediated I-PDT, a selected range of irradiance prompts effective photoreaction with tissue heating in the treatment of locally advanced mouse tumour. These irradiances were translated for effective local control of large VX2 tumours.
Conflict of interest statement
G.S., D.A.B., and E.O. are co-inventors in patent applications (owned by Roswell Park Comprehensive Cancer Center) of a light dosimetry system for interstitial and thermal photodynamic therapy. G.S. and H.A. acknowledge research grant support from Concordia Laboratories, Inc. G.S. acknowledges a service on the advisory board for Concordia International Corp. and Pinnacle Biologics, Inc. The remaining authors declare no competing interests. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or Roswell Park Comprehensive Cancer Center.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

