Mechanisms of Connexin-Related Lymphedema
- PMID: 30355030
- PMCID: PMC6771293
- DOI: 10.1161/CIRCRESAHA.117.312576
Mechanisms of Connexin-Related Lymphedema
Abstract
Rationale: Mutations in GJC2 and GJA1, encoding Cxs (connexins) 47 and 43, respectively, are linked to lymphedema, but the underlying mechanisms are unknown. Because efficient lymph transport relies on the coordinated contractions of lymphatic muscle cells (LMCs) and their electrical coupling through Cxs, Cx-related lymphedema is proposed to result from dyssynchronous contractions of lymphatic vessels.
Objective: To determine which Cx isoforms in LMCs and lymphatic endothelial cells are required for the entrainment of lymphatic contraction waves and efficient lymph transport.
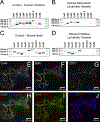
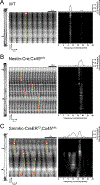
Methods and results: We developed novel methods to quantify the spatiotemporal entrainment of lymphatic contraction waves and used optogenetic techniques to analyze calcium signaling within and between the LMC and the lymphatic endothelial cell layers. Genetic deletion of the major lymphatic endothelial cell Cxs (Cx43, Cx47, or Cx37) revealed that none were necessary for the synchronization of the global calcium events that triggered propagating contraction waves. We identified Cx45 in human and mouse LMCs as the critical Cx mediating the conduction of pacemaking signals and entrained contractions. Smooth muscle-specific Cx45 deficiency resulted in 10- to 18-fold reduction in conduction speed, partial-to-severe loss of contractile coordination, and impaired lymph pump function ex vivo and in vivo. Cx45 deficiency resulted in profound inhibition of lymph transport in vivo, but only under an imposed gravitational load.
Conclusions: Our results (1) identify Cx45 as the Cx isoform mediating the entrainment of the contraction waves in LMCs; (2) show that major endothelial Cxs are dispensable for the entrainment of contractions; (3) reveal a lack of coupling between lymphatic endothelial cells and LMCs, in contrast to arterioles; (4) point to lymphatic valve defects, rather than contraction dyssynchrony, as the mechanism underlying GJC2- or GJA1-related lymphedema; and (5) show that a gravitational load exacerbates lymphatic contractile defects in the intact mouse hindlimb, which is likely critical for the development of lymphedema in the adult mouse.
Keywords: calcium signaling; connexins; lymph; lymphatic system; lymphedema; vascular disease.
Figures








References
-
- Ostergaard P, Simpson MA, Brice G, Mansour S, Connell FC, Onoufriadis A, Child AH, Hwang J, Kalidas K, Mortimer PS, Trembath R, Jeffery S. Rapid identification of mutations in gjc2 in primary lymphoedema using whole exome sequencing combined with linkage analysis with delineation of the phenotype. J Med Genet. 2011;48:251–255 - PubMed
-
- Finegold DN, Baty CJ, Knickelbein KZ, Perschke S, Noon SE, Campbell D, Karlsson JM, Huang D, Kimak MA, Lawrence EC, Feingold E, Meriney SD, Brufsky AM, Ferrell RE. Connexin 47 mutations increase risk for secondary lymphedema following breast cancer treatment. Clin Cancer Res. 2012;18:2382–2390 - PMC - PubMed
-
- Brice G, Ostergaard P, Jeffery S, Gordon K, Mortimer PS, Mansour S. A novel mutation in gja1 causing oculodentodigital syndrome and primary lymphoedema in a three generation family. Clin Genet. 2013;84:378–381 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

