5-HT2A Receptor-Induced Morphological Reorganization of PKCγ-Expressing Interneurons Gates Inflammatory Mechanical Allodynia in Rat
- PMID: 30355630
- PMCID: PMC6596258
- DOI: 10.1523/JNEUROSCI.1294-18.2018
5-HT2A Receptor-Induced Morphological Reorganization of PKCγ-Expressing Interneurons Gates Inflammatory Mechanical Allodynia in Rat
Abstract
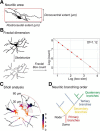
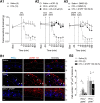
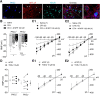
Mechanical allodynia, a widespread pain symptom that still lacks effective therapy, is associated with the activation of a dorsally directed polysynaptic circuit within the spinal dorsal horn (SDH) or medullary dorsal horn (MDH), whereby tactile inputs into deep SDH/MDH can gain access to superficial SDH/MDH, eliciting pain. Inner lamina II (IIi) interneurons expressing the γ isoform of protein kinase C (PKCγ+) are key elements for allodynia circuits, but how they operate is still unclear. Combining behavioral, ex vivo electrophysiological, and morphological approaches in an adult rat model of facial inflammatory pain (complete Freund's adjuvant, CFA), we show that the mechanical allodynia observed 1 h after CFA injection is associated with the following (1) sensitization (using ERK1/2 phosphorylation as a marker) and (2) reduced dendritic arborizations and enhanced spine density in exclusively PKCγ+ interneurons, but (3) depolarized resting membrane potential (RMP) in all lamina IIi PKCγ+/PKCγ- interneurons. Blocking MDH 5HT2A receptors (5-HT2AR) prevents facial mechanical allodynia and associated changes in the morphology of PKCγ+ interneurons, but not depolarized RMP in lamina IIi interneurons. Finally, activation of MDH 5-HT2AR in naive animals is enough to reproduce the behavioral allodynia and morphological changes in PKCγ+ interneurons, but not the electrophysiological changes in lamina IIi interneurons, induced by facial inflammation. This suggests that inflammation-induced mechanical allodynia involves strong morphological reorganization of PKCγ+ interneurons via 5-HT2AR activation that contributes to open the gate for transmission of innocuous mechanical inputs to superficial SDH/MDH pain circuitry. Preventing 5-HT2AR-induced structural plasticity in PKCγ+ interneurons might represent new avenues for the specific treatment of inflammation-induced mechanical hypersensitivity.SIGNIFICANCE STATEMENT Inflammatory or neuropathic pain syndromes are characterized by pain hypersensitivity such as mechanical allodynia (pain induced by innocuous mechanical stimuli). It is generally assumed that mechanisms underlying mechanical allodynia, because they are rapid, must operate at only the level of functional reorganization of spinal or medullary dorsal horn (MDH) circuits. We discovered that facial inflammation-induced mechanical allodynia is associated with rapid and strong structural remodeling of specifically interneurons expressing the γ isoform of protein kinase C (PKCγ) within MDH inner lamina II. Moreover, we elucidated a 5-HT2A receptor to PKCγ/ERK1/2 pathway leading to the behavioral allodynia and correlated morphological changes in PKCγ interneurons. Therefore, descending 5-HT sensitize PKCγ interneurons, a putative "gate" in allodynia circuits, via 5-HT2A receptor-induced structural reorganization.
Keywords: 5-HT2A; PKC-gamma; inflammation; medullary dorsal horn; pain; serotonin.
Copyright © 2018 the authors 0270-6474/18/3810490-16$15.00/0.
Figures





Comment in
-
Dorsal Horn PKCγ Interneurons Mediate Mechanical Allodynia through 5-HT2AR-Dependent Structural Reorganization.J Neurosci. 2019 Aug 7;39(32):6221-6223. doi: 10.1523/JNEUROSCI.0291-19.2019. J Neurosci. 2019. PMID: 31391259 Free PMC article. No abstract available.
References
-
- Aira Z, Buesa I, Salgueiro M, Bilbao J, Aguilera L, Zimmermann M, Azkue JJ (2010) Subtype-specific changes in 5-HT receptor-mediated modulation of C fibre-evoked spinal field potentials are triggered by peripheral nerve injury. Neuroscience 168:831–841. 10.1016/j.neuroscience.2010.04.032 - DOI - PubMed
-
- Aira Z, Buesa I, Gallego M, García del Caño G, Mendiable N, Mingo J, Rada D, Bilbao J, Zimmermann M, Azkue JJ (2012) Time-dependent cross talk between spinal serotonin 5-HT2A receptor and mGluR1 subserves spinal hyperexcitability and neuropathic pain after nerve injury. J Neurosci 32:13568–13581. 10.1523/JNEUROSCI.1364-12.2012 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
