Divergent changes of p53 in pulmonary arterial endothelial and smooth muscle cells involved in the development of pulmonary hypertension
- PMID: 30358436
- PMCID: PMC6383500
- DOI: 10.1152/ajplung.00538.2017
Divergent changes of p53 in pulmonary arterial endothelial and smooth muscle cells involved in the development of pulmonary hypertension
Abstract
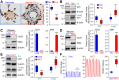
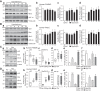
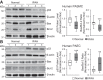
The tumor-suppressive role of p53, a transcription factor that regulates the expression of many genes, has been linked to cell cycle arrest, apoptosis, and senescence. The noncanonical function or the pathogenic role of p53 has more recently been implicated in pulmonary vascular disease. We previously reported that rapid nuclear accumulation of hypoxia-inducible factor (HIF)-1α in pulmonary arterial smooth muscle cells (PASMCs) upregulates transient receptor potential channels and enhances Ca2+ entry to increase cytosolic Ca2+ concentration ([Ca2+]cyt). Also, we observed differences in HIF-1α/2α expression in PASMCs and pulmonary arterial endothelial cells (PAECs). Here we report that p53 is increased in PAECs, but decreased in PASMCs, isolated from mice with hypoxia-induced pulmonary hypertension (PH) and rats with monocrotaline (MCT)-induced PH (MCT-PH). The increased p53 in PAECs from rats with MCT-PH is associated with an increased ratio of Bax/Bcl-2, while the decreased p53 in PASMCs is associated with an increased HIF-1α. Furthermore, p53 is downregulated in PASMCs isolated from patients with idiopathic pulmonary arterial hypertension compared with PASMCs from normal subjects. Overexpression of p53 in normal PASMCs inhibits store-operated Ca2+ entry (SOCE) induced by passive depletion of intracellularly stored Ca2+ in the sarcoplasmic reticulum, while downregulation of p53 enhances SOCE. These data indicate that differentially regulated expression of p53 and HIF-1α/2α in PASMCs and PAECs and the cross talk between p53 and HIF-1α/2α in PASMCs and PAECs may play an important role in the development of PH via, at least in part, induction of PAEC apoptosis and PASMC proliferation.
Keywords: Bcl-2 proteins; endothelial cell apoptosis; p53; smooth muscle cell proliferation; tumor-suppressor gene.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures




References
-
- Abid S, Houssaïni A, Mouraret N, Marcos E, Amsellem V, Wan F, Dubois-Randé JL, Derumeaux G, Boczkowski J, Motterlini R, Adnot S. P21-dependent protective effects of a carbon monoxide-releasing molecule-3 in pulmonary hypertension. Arterioscler Thromb Vasc Biol 34: 304–312, 2014. doi: 10.1161/ATVBAHA.113.302302. - DOI - PubMed
-
- Archer SL, Gomberg-Maitland M, Maitland ML, Rich S, Garcia JG, Weir EK. Mitochondrial metabolism, redox signaling, and fusion: a mitochondria-ROS-HIF-1α-Kv1.5 O2-sensing pathway at the intersection of pulmonary hypertension and cancer. Am J Physiol Heart Circ Physiol 294: H570–H578, 2008. doi: 10.1152/ajpheart.01324.2007. - DOI - PubMed
-
- Ball MK, Waypa GB, Mungai PT, Nielsen JM, Czech L, Dudley VJ, Beussink L, Dettman RW, Berkelhamer SK, Steinhorn RH, Shah SJ, Schumacker PT. Regulation of hypoxia-induced pulmonary hypertension by vascular smooth muscle hypoxia-inducible factor-1α. Am J Respir Crit Care Med 189: 314–324, 2014. doi: 10.1164/rccm.201302-0302OC. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

