Novel compounds that specifically bind and modulate MscL: insights into channel gating mechanisms
- PMID: 30359098
- PMCID: PMC6404570
- DOI: 10.1096/fj.201801628R
Novel compounds that specifically bind and modulate MscL: insights into channel gating mechanisms
Abstract
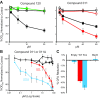
The bacterial mechanosensitive channel of large conductance (MscL) normally functions as an emergency release valve discharging cytoplasmic solutes upon osmotic stress. Opening the large pore of MscL inappropriately is detrimental to the cell, and thus it has been speculated to be a potential antibiotic target. Although MscL is one of the best studied mechanosensitive channels, no chemical that influenced bacterial growth by modulating MscL is known. We therefore used a high-throughput screen to identify compounds that slowed growth in an MscL-dependent manner. We characterized 2 novel sulfonamide compounds identified in the screen. We demonstrated that, although both increase MscL gating, one of these compounds does not work through the folate pathway, as other antimicrobial sulfonamides; indeed, the sulfonamide portion of the compound is not needed for activity. The only mode of action appears to be MscL activation. The binding pocket is where an α-helix runs along the cytoplasmic membrane and interacts with a neighboring subunit; analogous motifs have been observed in several prokaryotic and eukaryotic channels. The data not only demonstrate that MscL is a viable antibiotic target, but also give insight into the gating mechanisms of MscL, and they may have implications for developing agonists for other channels.-Wray, R., Iscla, I., Kovacs, Z., Wang, J., Blount, P. Novel compounds that specifically bind and modulate MscL: insights into channel gating mechanisms.
Keywords: antibiotic; mechanosensitive; osmoregulation.
Conflict of interest statement
The authors thank Drs. Limin Yang (University of Texas, Southwestern Medical Center) and Andriy Anishkin (University of Maryland, College Park, MD, USA) for helpful suggestions and critical reading of the manuscript. This work was supported by U.S. National Institutes of Health (NIH) National Institute of General Medical Sciences (NIGMS) Grants R01 GM061028 and GM121780, and NIH National Institute of Biomedical Imaging and Bioengineering Grant P41-EB015908 (to Z.K.); Welch Foundation Grant I-1420 (to P.B.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or other funding organizations. The authors declare no conflicts of interest.
Figures



References
-
- Steinbacher S., Bass R., Strop P., Rees D. C. (2007) Structures of the prokaryotic mechanosensitive channels MscL and MscS: mechanosensitive ion channels. Curr Topics Membranes. 58, 1–20,
-
- Cox C. D., Bavi N., Martinac B. (2018) Bacterial mechanosensors. Annu. Rev. Physiol. 80, 71–93 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

